Chapter 4: Q.47. (page 128)
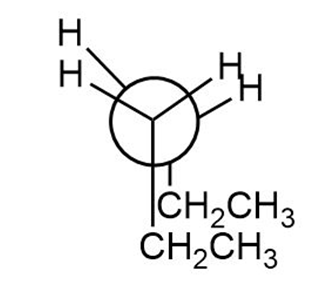
Question: Convert each structure to a Newman projection around the bond highlighted in pink.
a.
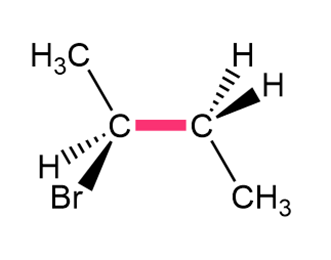
b.
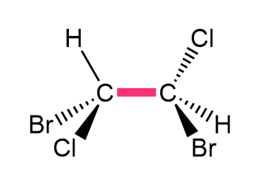
c.
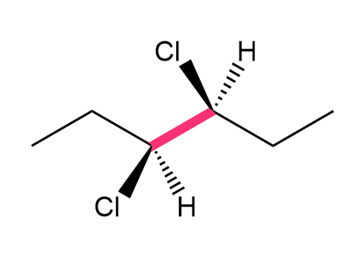
Short Answer
Answer
a.
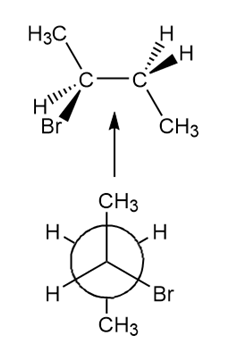
b.
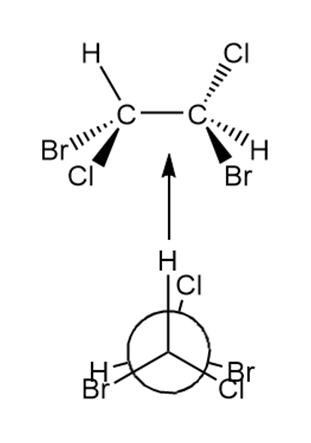
c.
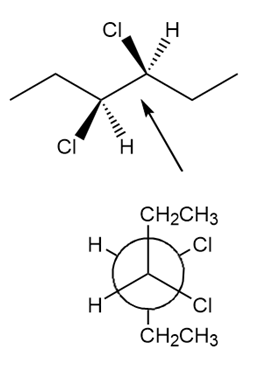
Learning Materials
Features
Discover
Chapter 4: Q.47. (page 128)
Question: Convert each structure to a Newman projection around the bond highlighted in pink.
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
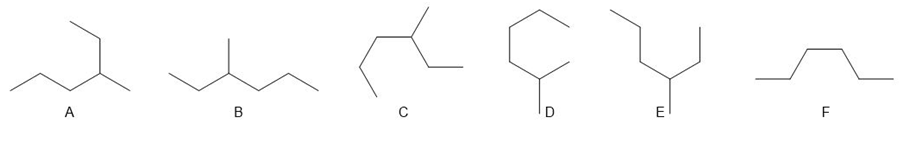
Question: a. Which compounds (B–F) are identical to A? b. Which compounds (B–F) represent an isomer of A?
Question: Draw both conformations for 1-ethyl-1-methylcyclohexane and decide which conformation (if any) is more stable.
Question: Convert each three-dimensional model to a Newman projection around the indicated bond.
Question: Label each compound as cis or trans. Then draw the second chair conformation.
a.
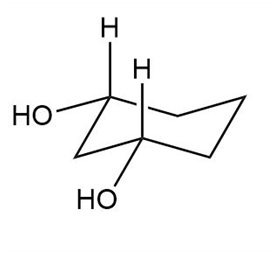
b.
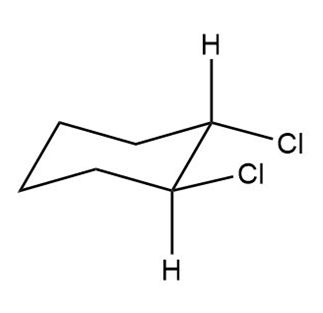
c.
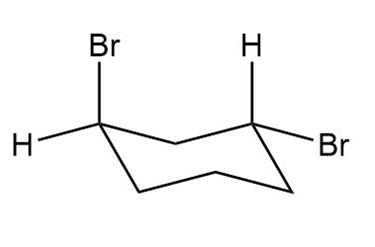
Question: Label the sites of torsional and steric strain in each conformation.
a.
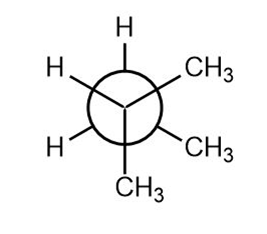
b.
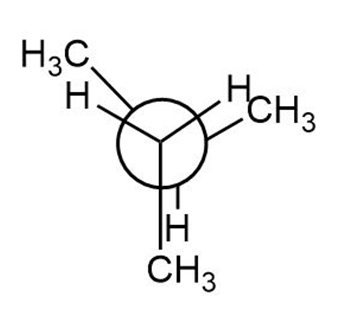
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.