Chapter 4: Q.36. (page 128)
Question: Convert each three-dimensional model to a Newman projection around the indicated bond.
Short Answer
Answer
a.
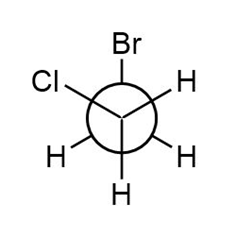
Newman Projection
b.
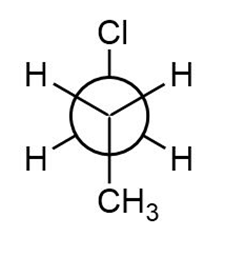
Newman projection
c.
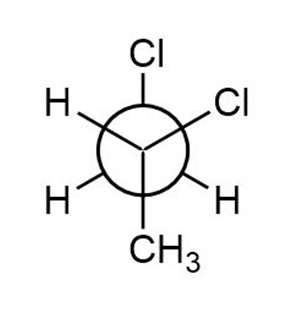
Newman projection
Learning Materials
Features
Discover
Chapter 4: Q.36. (page 128)
Question: Convert each three-dimensional model to a Newman projection around the indicated bond.
Answer
a.

Newman Projection
b.

Newman projection
c.

Newman projection
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the five constitutional isomers having molecular formula .
Question: Using the cyclohexane with the C’s numbered as shown, draw a chair form that fits each description.
a. The ring has an axial group at C1 and an equatorial OH on C2.
b. The ring has an equatorial group on C6 and an axial OH group on C4.
c. The ring has equatorial OH groups on C1, C2, and C5.
Question: Mineral oil, a mixture of high molecular weight alkanes, is sometimes used as a laxative. Why are individuals who use mineral oil for this purpose advised to avoid taking it at the same time they consume foods rich in fat-soluble vitamins such as vitamin A?
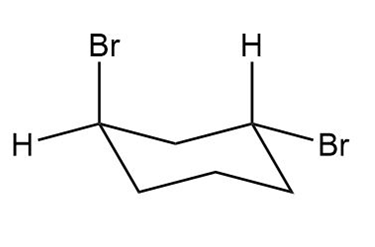
Question: Label each compound as cis or trans. Then draw the second chair conformation.
a.
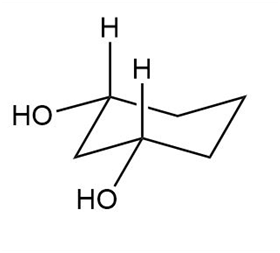
b.
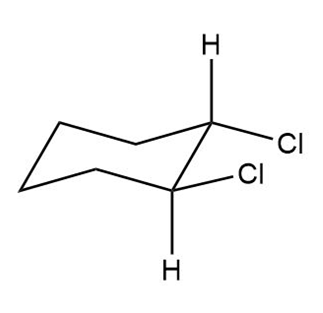
c.
Question: Each of the following IUPAC names is incorrect. Explain why it is incorrect and give the correct IUPAC name.
a. 2,2-dimethyl-4-ethylheptane
b. 5-ethyl-2-methylhexane
c. 2-methyl-2-isopropylheptane
d. 1,5-dimethylcyclohexane
e. 1-ethyl-2,6-dimethylcycloheptane
f. 5,5,6-trimethyloctane
g. 3-butyl-2,2-dimethylhexaneh. 1,3-dimethylbutane
What do you think about this solution?
We value your feedback to improve our textbook solutions.