To identify the cis and trans isomers, the positions of the functional groups and the hydrogen atoms have to be categorized as axial and equatorial positions.
The groups on the same side, i.e., axial or equatorial, represent the cis isomers. The groups on the opposite sides are categorized as trans-isomers.
a. The given compound is a cis isomer. Both hydroxyl groups are present at the equatorial positions. The hydroxyl groups are present as down bonds, which mean both are below the plane.
The hydrogen atoms present on the same carbon of the hydroxy groups are positioned upward, i.e., above the plane as shown in the structure below:
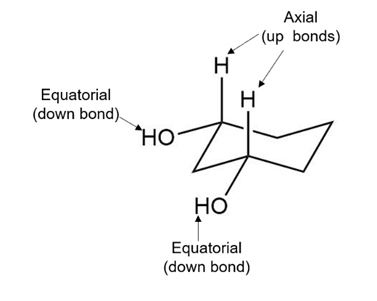
Cis cyclohexan-1,3-diol
The second chair conformation can be obtained by the flipping of the ring. It is shown as follows:
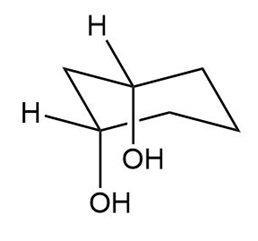
Cis cyclohexan-1,3-diol
b. The given compound is a trans isomer. In this compound, both chlorine atoms lie in the same plane.
At one position, as shown in the structure, the chlorine atom is present at the down position, while it is present at the upward position. Both these positions of the chlorine atom are equatorial, as shown in the structures below:
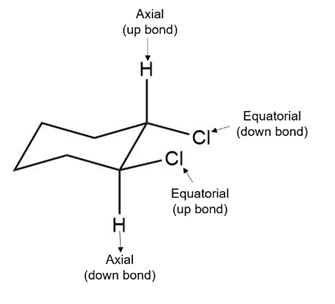
Trans- 1,2-dichloro cyclohexane
The second chair conformation can be obtained by the flipping of the ring as represented in the following structure:
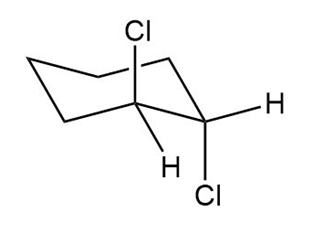
Trans-1,3-dichloro cyclohexane
c. The given compound is a trans isomer. One of the bromine atoms in this compound is present on the up bond, i.e., at the axial position.
The other bromine atom is present on the opposite side as the down bond, i.e., the equatorial position. The structure of the trans compound is shown below:
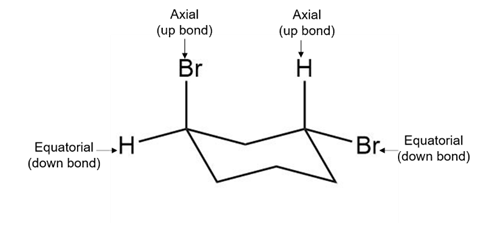
Trans- 1,3-dibromocyclohexane
The other isomer can be obtained by the flipping of the ring a shown below:
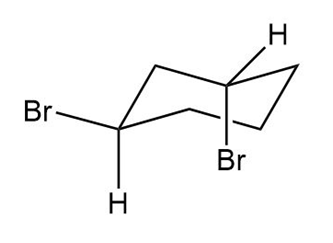
Trans-1,3 dibromo cyclohexane
















