Chapter 4: Q.26. (page 128)
Question: For cis-1,3-diethylcyclobutane, draw (a) a stereoisomer; (b) a constitutional isomer.
Short Answer
Answer
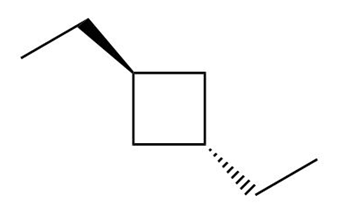
a. trans-1,3-diethylcyclobutane
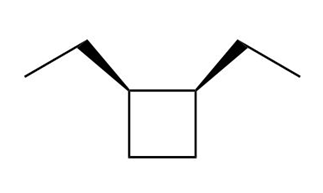
b. Cis-1,2-diethylcyclobutane
Learning Materials
Features
Discover
Chapter 4: Q.26. (page 128)
Question: For cis-1,3-diethylcyclobutane, draw (a) a stereoisomer; (b) a constitutional isomer.
Answer

a. trans-1,3-diethylcyclobutane

b. Cis-1,2-diethylcyclobutane
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The eclipsed conformation of is role="math" localid="1648620042367" less stable than the staggered conformation. How much is the H, Cl eclipsing interaction worth in destabilization?
Question: Convert each three-dimensional model to a Newman projection around the indicated bond.
Question: Read Appendix B on naming branched alkyl substituents, and draw all possible alkyl groups having the formula . Give the IUPAC names for the eight compounds of molecular formula that contain a cyclopentane ring with each of these alkyl groups as a substituent.
Question:Give IUPAC name of each compound.
a.
b.
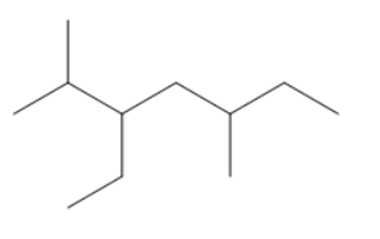
c.
d.
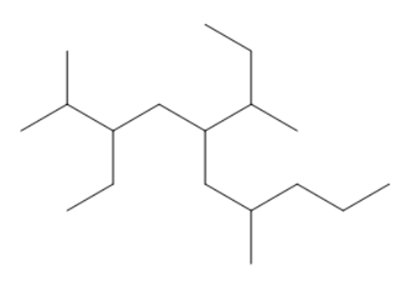
Question: Which conformation in each pair is higher in energy? Calculate the energy difference between the two conformations using the values given in Table 4.3.
a.
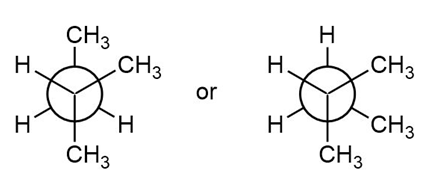
b.
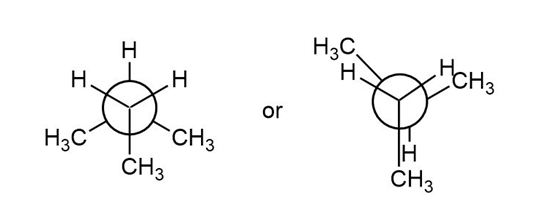
What do you think about this solution?
We value your feedback to improve our textbook solutions.