Chapter 4: Q.3. (page 128)
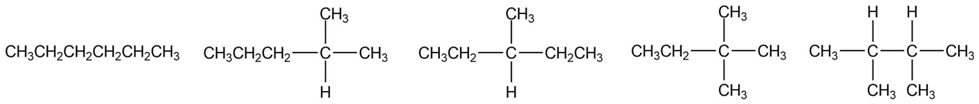
Question: Draw the five constitutional isomers having molecular formula .
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 4: Q.3. (page 128)
Question: Draw the five constitutional isomers having molecular formula .
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Each of the following IUPAC names is incorrect. Explain why it is incorrect and give the correct IUPAC name.
a. 2,2-dimethyl-4-ethylheptane
b. 5-ethyl-2-methylhexane
c. 2-methyl-2-isopropylheptane
d. 1,5-dimethylcyclohexane
e. 1-ethyl-2,6-dimethylcycloheptane
f. 5,5,6-trimethyloctane
g. 3-butyl-2,2-dimethylhexaneh. 1,3-dimethylbutane
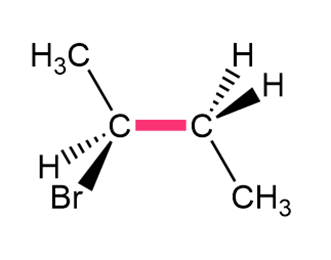
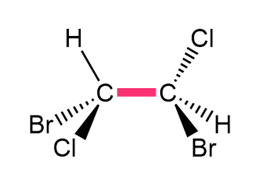
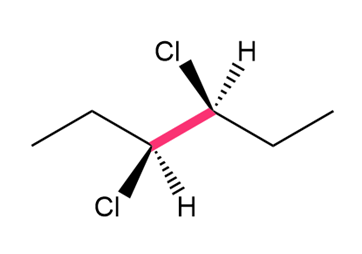
Question: Convert each structure to a Newman projection around the bond highlighted in pink.
a.
b.
c.
Question: Considering rotation around the bond highlighted in pink in each compound, draw Newman projections for the most stable and least stable conformations.
a.
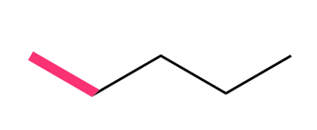
b.
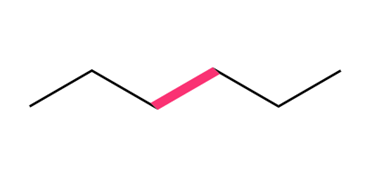
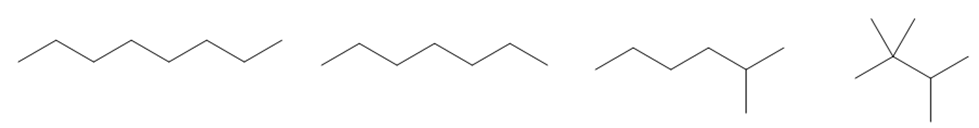
Question: Arrange the following compounds in order of increasing boiling point.
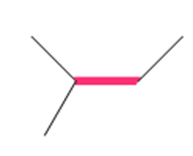
Question: a.Draw the three staggered and three eclipsed conformations that result from rotation around the bond labeled in red using Newman projections. b. Label the most stable and least stable conformation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.