Chapter 2: 2.27 (page 78)
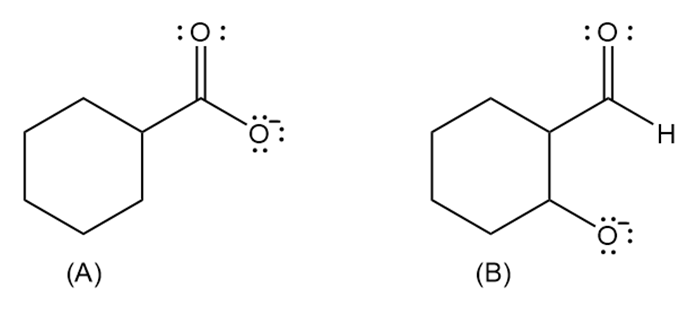
Which anion (A or B) is the stronger base?
Short Answer
B is the stronger base.
Learning Materials
Features
Discover
Chapter 2: 2.27 (page 78)
Which anion (A or B) is the stronger base?

B is the stronger base.
All the tools & learning materials you need for study success - in one app.
Get started for free
Molecules like acetamide can be protonated on either their O or N atoms when treated with a strong acid like HCl. Which site is more readily protonated and why?
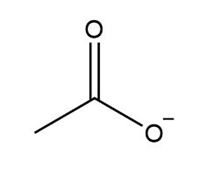
What is the conjugate acid of each base?
a.
b.
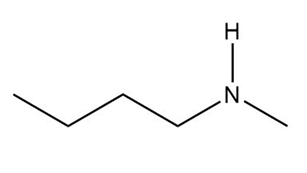
c.
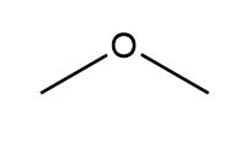
d.
Label the acid and base and the conjugate acid and base in the following reactions. Use curved arrows to show the movement of electron pairs.
a.
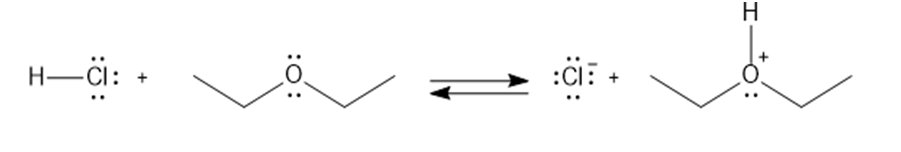
b.
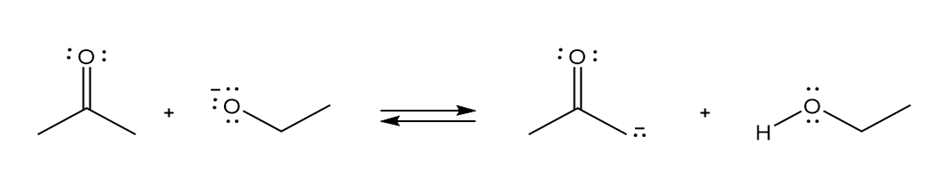
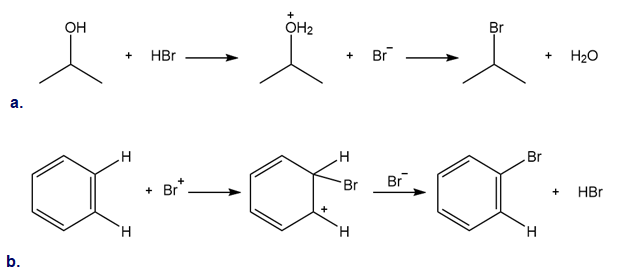
Classify each reaction as either a proton transfer reaction, or a reaction of a nucleophile with an electrophile. Use curved arrows to show how the electron pairs move.
Consider two acids: (formic acid, =3.8)and pivalic acid .
(a)Which acid has the larger ? (b)Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base?(d)When acid is dissolved in water, for which acid does the equilibrium lie further to the right?
What do you think about this solution?
We value your feedback to improve our textbook solutions.