Chapter 2: 2.28 (page 80)
Draw the products formed when propan-2-ol the main ingredient in rubbing alcohol, is treated with each acid or base: a. NaH b. c. d.
Short Answer
a.

b.
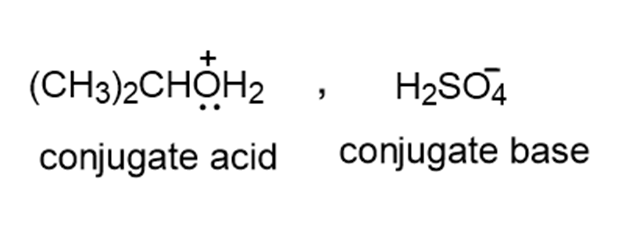
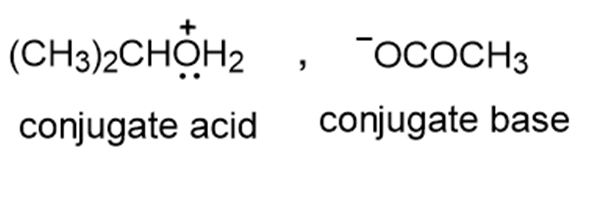
c.

d.
Learning Materials
Features
Discover
Chapter 2: 2.28 (page 80)
Draw the products formed when propan-2-ol the main ingredient in rubbing alcohol, is treated with each acid or base: a. NaH b. c. d.
a.

b.
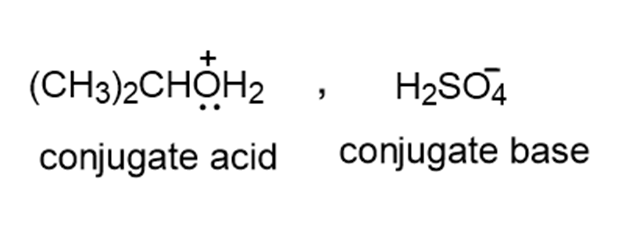
c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Two values are reported for malonic acid, a compound with two COOH groups. Explain why one is lower and one is higher than the of acetic acid

Bupivacaine (trade name Marcaine) is a quick-acting anesthetic often used during labor and delivery. Which nitrogen atom in bupivacaine is more basic? Explain your reasoning.

Answer the following questions about esmolol, a drug used to treat high blood pressure sold under the trade name Brevibloc.
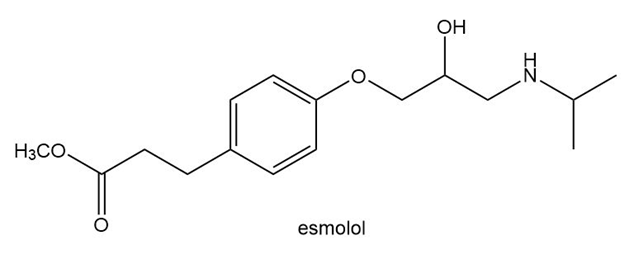
a. Label the most acidic hydrogen atom
b. What products are formed when esmolol is treated with NaH?
c. What products are formed when esmolol is treated with HCl?
d. Label all hybridized C atoms. in esmolol.
e. Label the only trigonal pyramidal atom.
f. Label all C’s that bear a charge.
Which of the following bases are strong enough to deprotonate ( = 25), so that equilibrium favors the products:
Explain the apparent paradox. HBr is a stronger acid than HCl, but HOCl is a stronger acid than HOBr.
What do you think about this solution?
We value your feedback to improve our textbook solutions.