Chapter 2: 2.21 (page 74)
Explain the apparent paradox. HBr is a stronger acid than HCl, but HOCl is a stronger acid than HOBr.
Short Answer
HOCl is a stronger acid than HOBr as the electronegativity of chlorine is higher than that of bromine.
Learning Materials
Features
Discover
Chapter 2: 2.21 (page 74)
Explain the apparent paradox. HBr is a stronger acid than HCl, but HOCl is a stronger acid than HOBr.
HOCl is a stronger acid than HOBr as the electronegativity of chlorine is higher than that of bromine.
All the tools & learning materials you need for study success - in one app.
Get started for free
Classify each reaction as either a proton transfer reaction, or a reaction of a nucleophile with an electrophile. Use curved arrows to show how the electron pairs move.
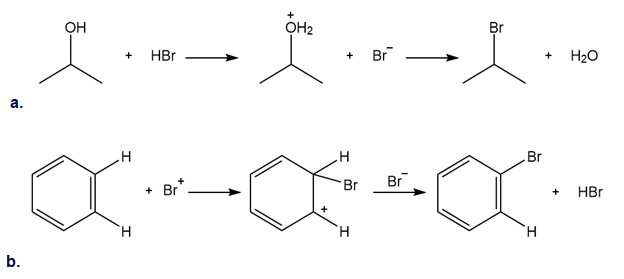
a. Draw the conjugate acid of each base:
b.Draw the conjugate base of each acid: HBr,
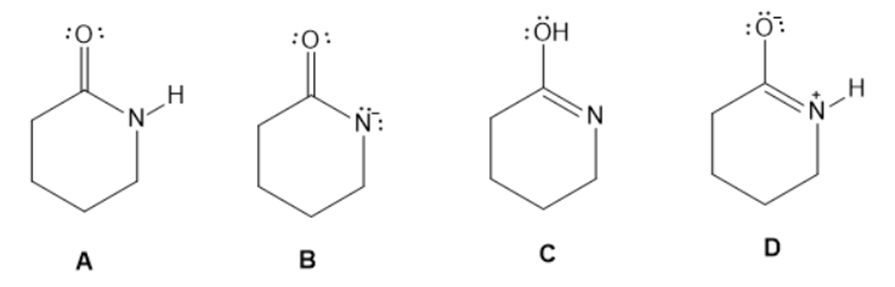
Answer the following questions about the four species A-D.
a. Which two species represent a conjugate acid-base pair?
b. Which two species represent resonance structures?
c. Which two species represent constitutional isomers?
Label each statement as True or False.
a.is the conjugate acid of.
b.is the conjugate base of.
c.is the conjugate base of.
d.is the conjugate base of.
e. is the conjugate acid of .
Which hydrogen in each molecule is most acidic?
a.
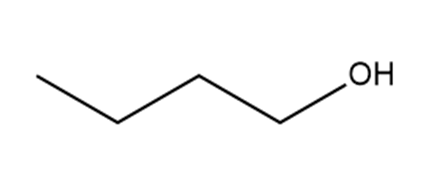
b.
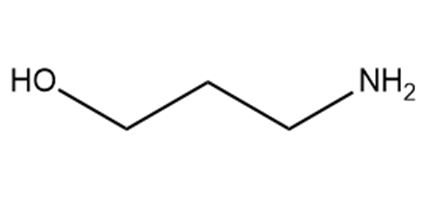
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.