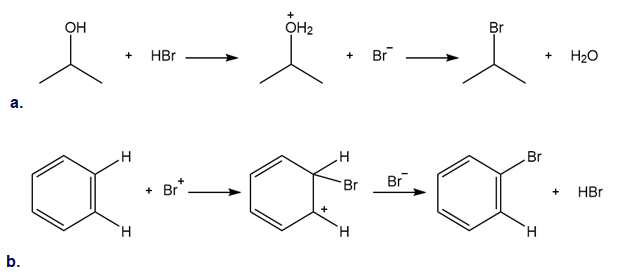
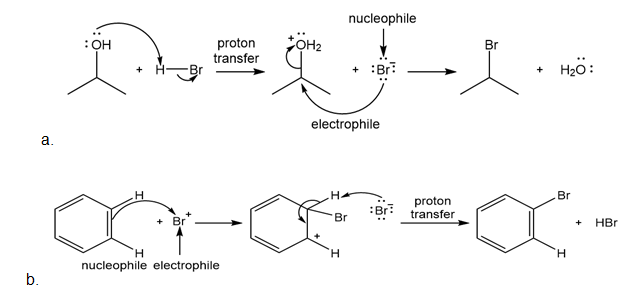
Chapter 2: Q. 2.69 (page 89)
Classify each reaction as either a proton transfer reaction, or a reaction of a nucleophile with an electrophile. Use curved arrows to show how the electron pairs move.
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q. 2.69 (page 89)
Classify each reaction as either a proton transfer reaction, or a reaction of a nucleophile with an electrophile. Use curved arrows to show how the electron pairs move.


All the tools & learning materials you need for study success - in one app.
Get started for free
Dimethyl ether and ethanol are isomers, but has a role="math" localid="1649252571433" of 40 and has a of 16. Why are these values so different?
Question: Use a calculator when necessary to answer the following questions.
a. What is the for each : ,and ?
b. What is the for each : 7, 11, and 3.2?
Amphetamine is a powerful stimulant of the central nervous system.
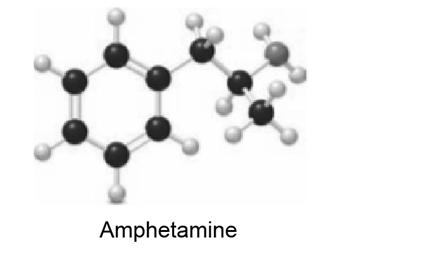
a. Which proton in amphetamine is most acidic? b. What products are formed when amphetamine is treated with NaH? c. What products are formed when amphetamine is treated with HCl?
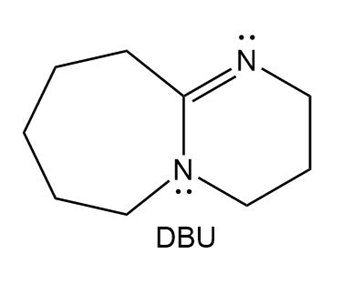
DBU, 1,8-diazabicyclo undec-7-ene, is a base we will encounter in elimination reactions in Chapter 8. Which N atom is more basic in DBU? Explain your choice.
Acetonitrile has a of 25, making it more acidic than many other compounds having only C-H bonds. Draw Lewis structures for acetonitrile and its conjugate base. Use resonance structures to account for the acidity of acetonitrile.
What do you think about this solution?
We value your feedback to improve our textbook solutions.