Chapter 2: 2.16 (page 72)
Which hydrogen in each molecule is most acidic?
a.
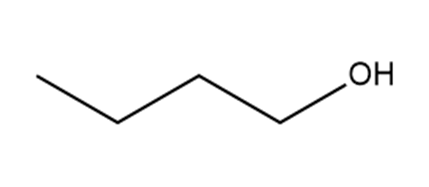
b.
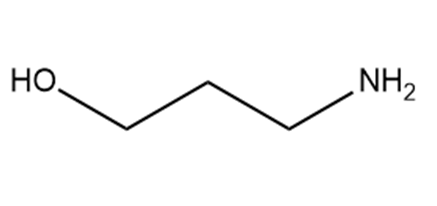
c.
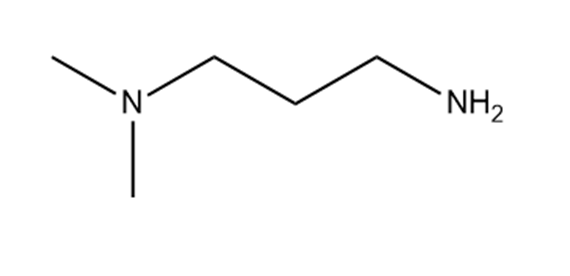
Short Answer
a.
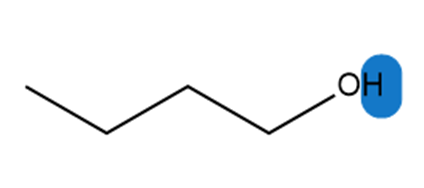
b.
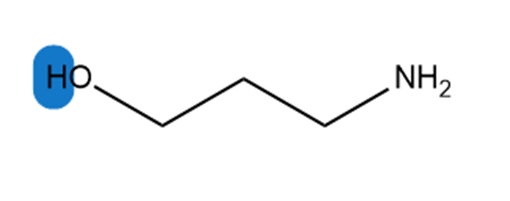
c.
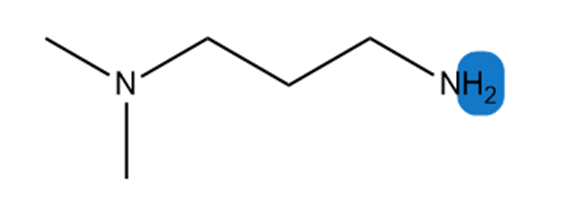
Learning Materials
Features
Discover
Chapter 2: 2.16 (page 72)
Which hydrogen in each molecule is most acidic?
a.

b.

c.

a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
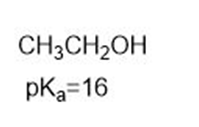
Which compound in each pair is the strongest acid?
a.
 or
or
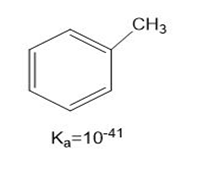
b.
 or
or
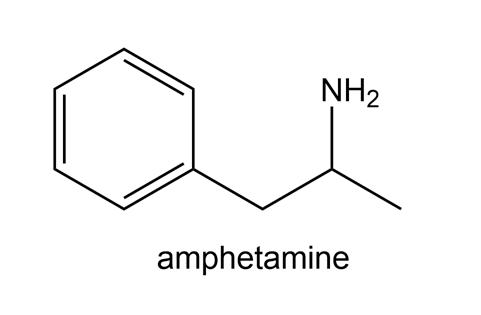
Compounds like amphetamine that contain nitrogen atoms are protonated by the HCl in the gastric juices of the stomach, and the resulting salt is then deprotonated in the basic environment of the intestines to regenerate the neutral form. Write proton transfer reactions for both of these processes. In which form will amphetamine pass through a cell membrane?
Dimethyl ether and ethanol are isomers, but has a role="math" localid="1649252571433" of 40 and has a of 16. Why are these values so different?
Consider two acids: (formic acid, =3.8)and pivalic acid .
(a)Which acid has the larger ? (b)Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base?(d)When acid is dissolved in water, for which acid does the equilibrium lie further to the right?
Draw the products of each proton transfer reaction. Label the acid and base in the starting materials, and the conjugate acid and base in the products.
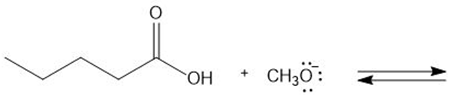
a.
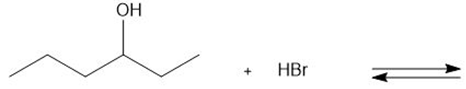
b.

c.
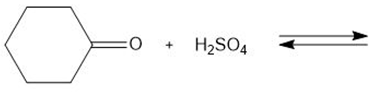
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.