Chapter 2: Q. 42 (page 85)
Draw the products of each proton transfer reaction. Label the acid and base in the starting materials, and the conjugate acid and base in the products.
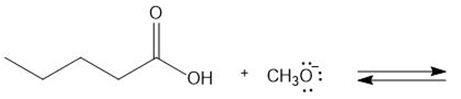
a.
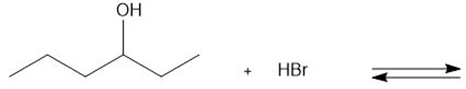
b.

c.
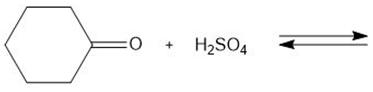
d.
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q. 42 (page 85)
Draw the products of each proton transfer reaction. Label the acid and base in the starting materials, and the conjugate acid and base in the products.

a.
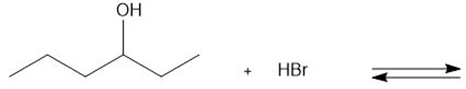
b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
a. Draw the conjugate acid of each base:
b.Draw the conjugate base of each acid: HBr,
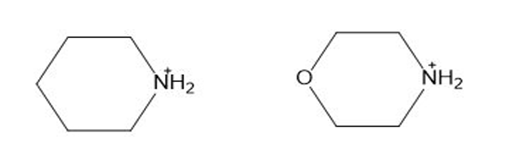
The values of the two ammonium cations drawn below are 8.33 and 11.1. Which corresponds to which cation? Explain your choice.
Draw the product formed when the Lewis acid reacts with each Lewis base:
role="math" localid="1649304251552"
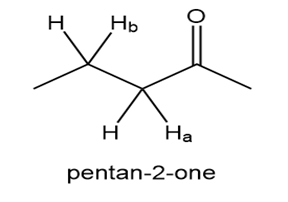
Explain why the bond is much more acidic than the bond in pentan-2-one.
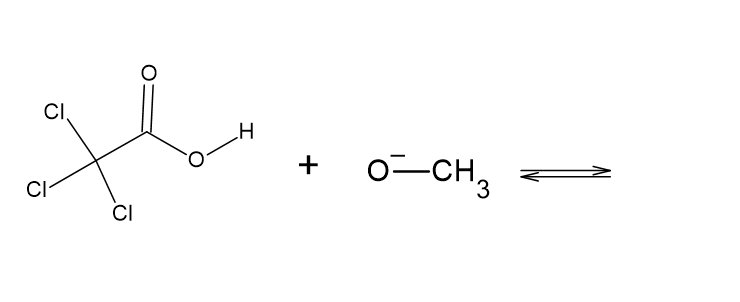
Decide which compound is the acid and which is the base, and draw the products of each proton transfer reaction.
(a)
(b)
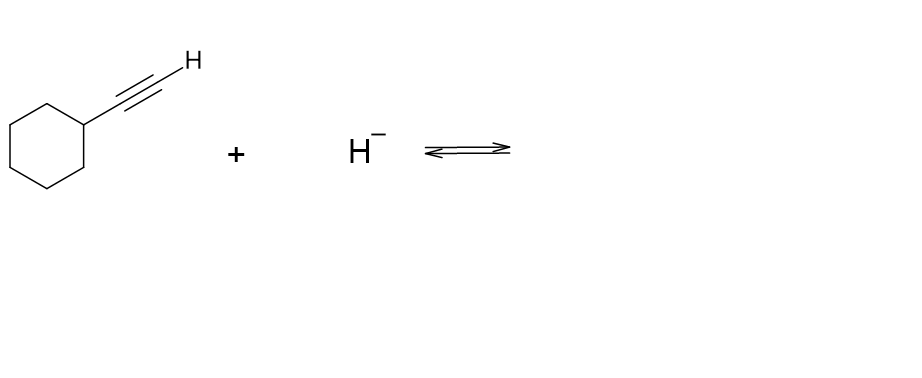
(c)
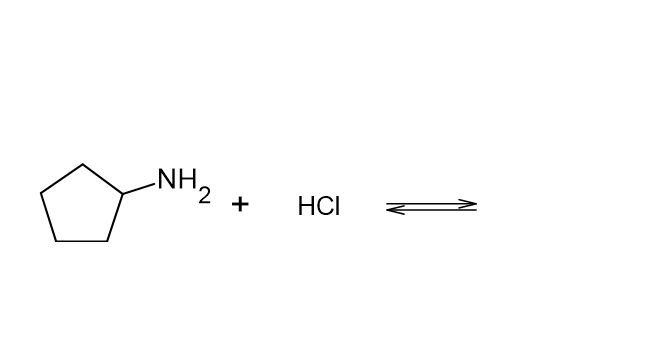
(d)
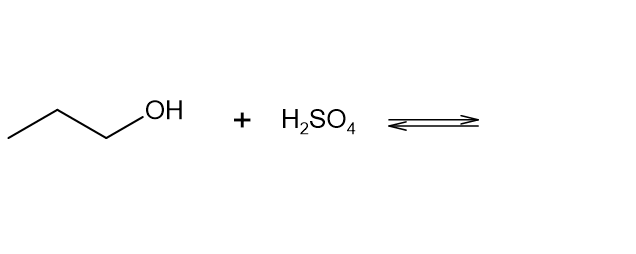
What do you think about this solution?
We value your feedback to improve our textbook solutions.