Chapter 2: Q. 2.53 (page 87)
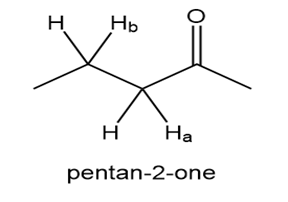
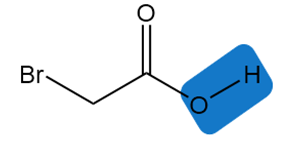
Explain why the bond is much more acidic than the bond in pentan-2-one.
Short Answer
The resonance stabilization makes to be more acidic than .
Learning Materials
Features
Discover
Chapter 2: Q. 2.53 (page 87)
Explain why the bond is much more acidic than the bond in pentan-2-one.

The resonance stabilization makes to be more acidic than .
All the tools & learning materials you need for study success - in one app.
Get started for free
Propranolol is an antihypertensive agent—that is, it lowers blood pressure.
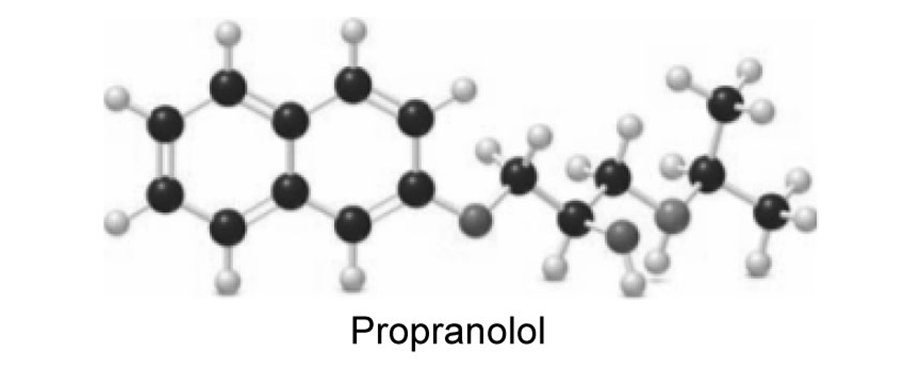
a. Which proton in propranolol is most acidic? b.What products are formed when propranolol is treated with NaH? c. Which atom is most basic? d. What products are formed when propranolol is treated with HCl?
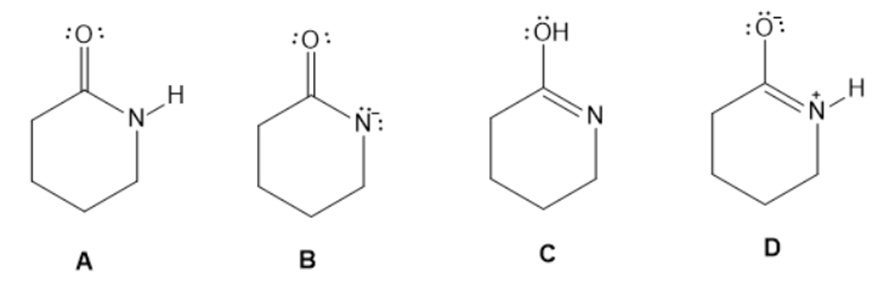
Answer the following questions about the four species A-D.
a. Which two species represent a conjugate acid-base pair?
b. Which two species represent resonance structures?
c. Which two species represent constitutional isomers?
Molecules like acetamide can be protonated on either their O or N atoms when treated with a strong acid like HCl. Which site is more readily protonated and why?
Explain the apparent paradox. HBr is a stronger acid than HCl, but HOCl is a stronger acid than HOBr.
Estimate the of each of the indicated bonds.
a.
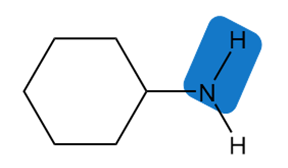
b.

c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.