Chapter 2: 2.11 (page 68)
Estimate the of each of the indicated bonds.
a.
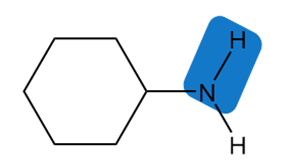
b.
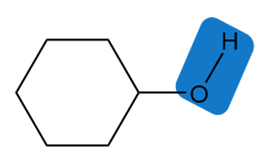
c.
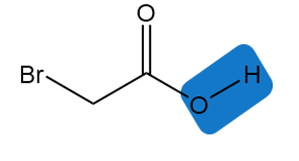
Short Answer
a. 38
b. 16
c.2.9
Learning Materials
Features
Discover
Chapter 2: 2.11 (page 68)
Estimate the of each of the indicated bonds.
a.

b.

c.

a. 38
b. 16
c.2.9
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider two acids: (formic acid, =3.8)and pivalic acid .
(a)Which acid has the larger ? (b)Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base?(d)When acid is dissolved in water, for which acid does the equilibrium lie further to the right?
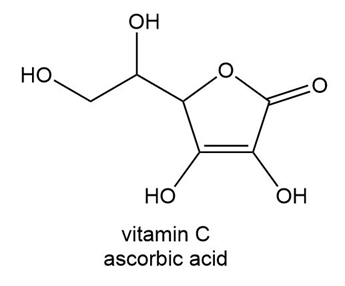
Which H atom in vitamin C (ascorbic acid) is most acidic?
Which anion (A or B) is the stronger base?
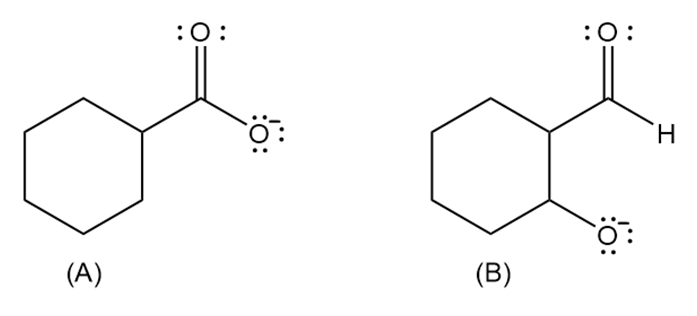
Draw the product formed when , a Lewis base, reacts with each Lewis acid:
a. role="math" localid="1648902828598"
b. role="math" localid="1648902808294"
c.
Use the principles in Section 2.5 to label the most acidic hydrogen in each drug. Explain your choice.
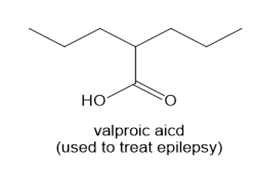
a.
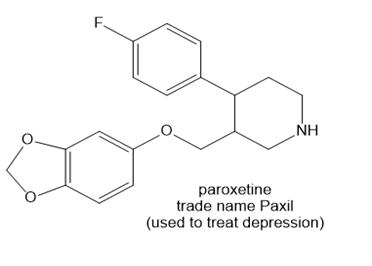
b.
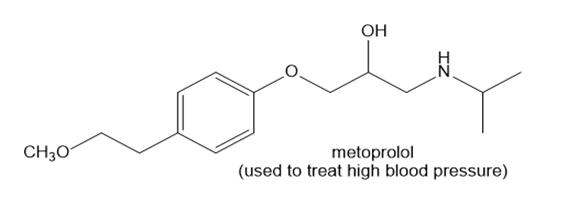
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.