Chapter 2: Q4. (page 61)
Question: Draw additional resonance structures for each anion.
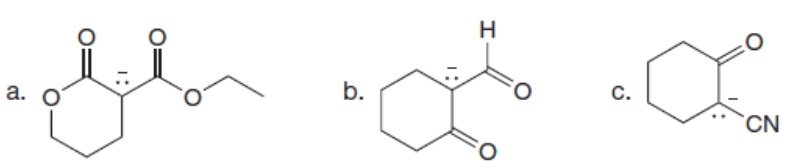
Short Answer
Answer
The additional resonance structure for each anion is shown below.
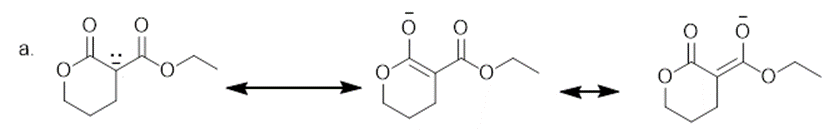
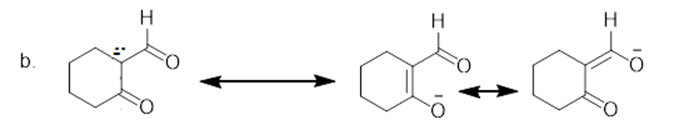
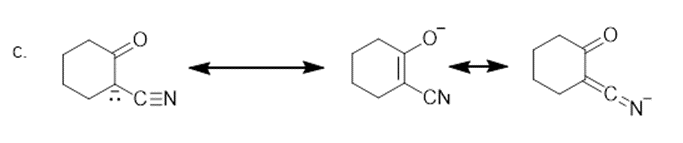
Learning Materials
Features
Discover
Chapter 2: Q4. (page 61)
Question: Draw additional resonance structures for each anion.

Answer
The additional resonance structure for each anion is shown below.



All the tools & learning materials you need for study success - in one app.
Get started for free
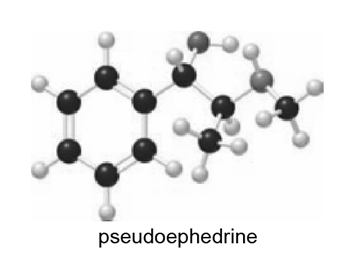
Which hydrogen in pseudoephedrine, the nasal decongestant in the commercial medication Sudafed, is most acidic?
Amino acids such as glycine are the building blocks of large molecules called proteins that give structure to muscle, tendon, hair, and nails.
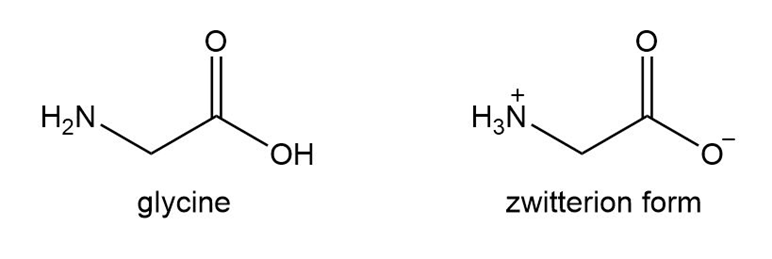
a. Explain why glycine does not actually exist in the form with all atoms uncharged, but actually exists as a salt called a zwitterion.
b. What product is formed when glycine is treated with concentrated HCl?
c. What product is formed when glycine is treated with NaOH?
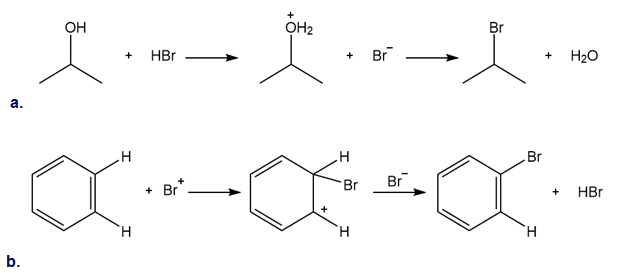
Classify each reaction as either a proton transfer reaction, or a reaction of a nucleophile with an electrophile. Use curved arrows to show how the electron pairs move.
Which compound in each pair is the strongest acid?
a.
 or
or
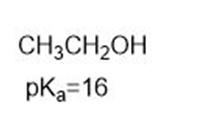
b.
 or
or
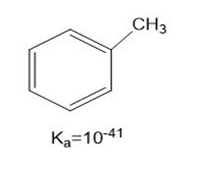
Which species are Lewis bases?
a.
b.
c.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.