Chapter 2: 2.30 (page 82)
Which species are Lewis bases?
a.
b.
c.
d.
Short Answer
a. Lewis base.
b. Not Lewis base.
c. Lewis base.
d. Lewis base.
Learning Materials
Features
Discover
Chapter 2: 2.30 (page 82)
Which species are Lewis bases?
a.
b.
c.
d.
a. Lewis base.
b. Not Lewis base.
c. Lewis base.
d. Lewis base.
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Draw the conjugate acid of each base:
b.Draw the conjugate base of each acid: HBr,
What is the conjugate acid of each base?
a.
b.
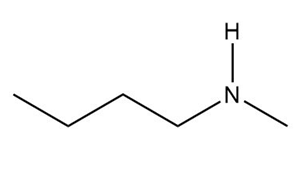
c.
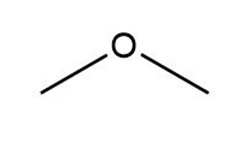
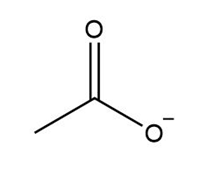
d.
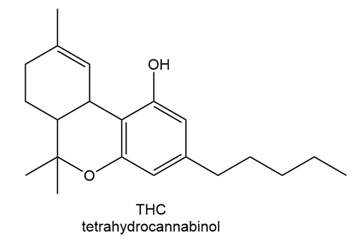
Which proton in each of the following drugs is most acidic? THC is the active component in marijuana, and ketoprofen is an anti-inflammatory agent.
a.
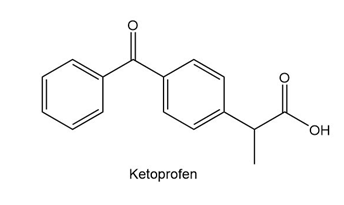
b.
Which compounds can be deprotonated by , so that equilibrium favors the products? Refer to the pKa table in Appendix A.
a.HCOOH
b.
c.
d.
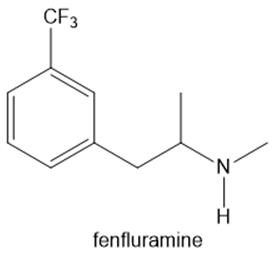
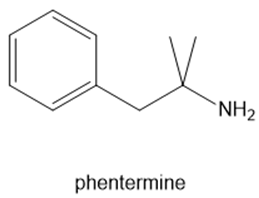
Fenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.