Chapter 2: 2.31 (page 82)
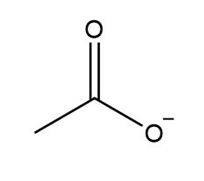
Which species are Lewis acids?
a.
b.
c.
d.role="math" localid="1648897351962"
Short Answer
a. Lewis acid
b. It is notLewis acid
c. Lewis acid
d. It is not a Lewis acid
Learning Materials
Features
Discover
Chapter 2: 2.31 (page 82)
Which species are Lewis acids?
a.
b.
c.
d.role="math" localid="1648897351962"
a. Lewis acid
b. It is notLewis acid
c. Lewis acid
d. It is not a Lewis acid
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Which compounds are Brønsted-Lowry acids: ?
b.Which compounds are Brønsted-Lowry bases:?
c. Classify each compound as an acid, a base, or both:
Draw the products formed from the acid–base reaction of with each compound.
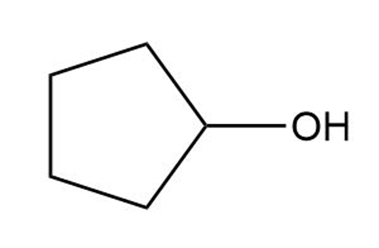
a.

b.
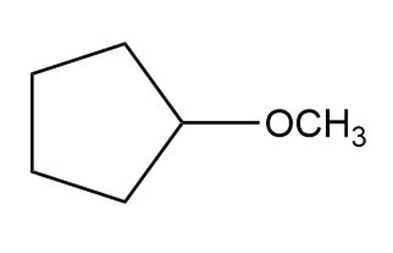
c.
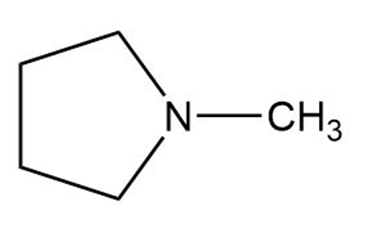
d.
Write a stepwise reaction sequence using proton transfer reactions to show how the following reaction occurs. (Hint: As a first step, use to remove a proton from the group between the C=O and C=C.)
Molecules like acetamide can be protonated on either their O or N atoms when treated with a strong acid like HCl. Which site is more readily protonated and why?
What is the conjugate acid of each base?
a.
b.
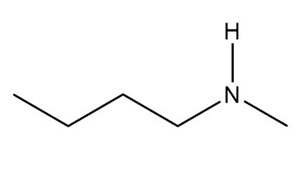
c.

d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.