Chapter 2: Q. 2.40 (page 85)
Draw the products formed from the acid–base reaction of with each compound.
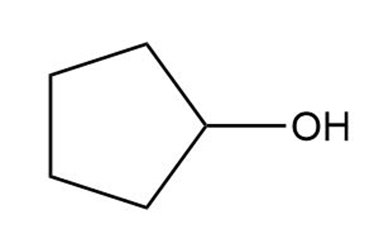
a.
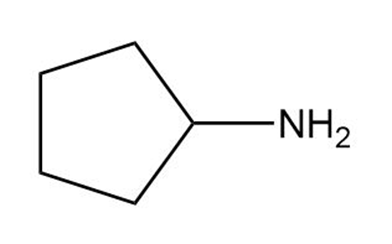
b.
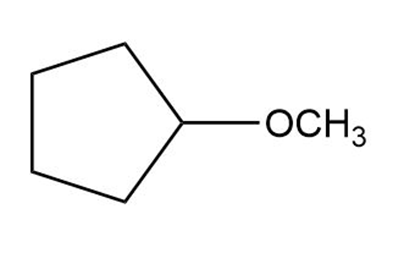
c.
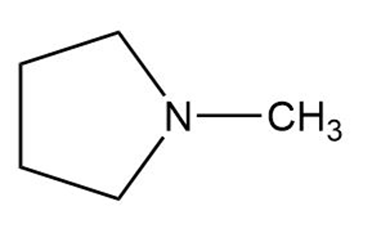
d.
Short Answer

Learning Materials
Features
Discover
Chapter 2: Q. 2.40 (page 85)
Draw the products formed from the acid–base reaction of with each compound.

a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
a. Draw the conjugate acid of each base:
b.Draw the conjugate base of each acid: HBr,
Draw the products formed when propan-2-ol the main ingredient in rubbing alcohol, is treated with each acid or base: a. NaH b. c. d.
Which of the following bases are strong enough to deprotonate ( = 25), so that equilibrium favors the products:
What is the for each compound?
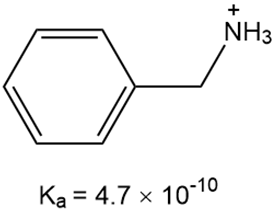
a.
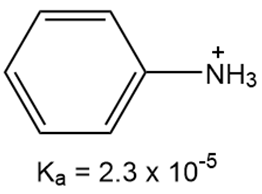
b.
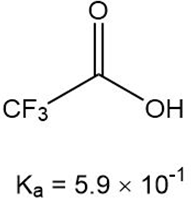
c.
The of three C-H bonds is given below.
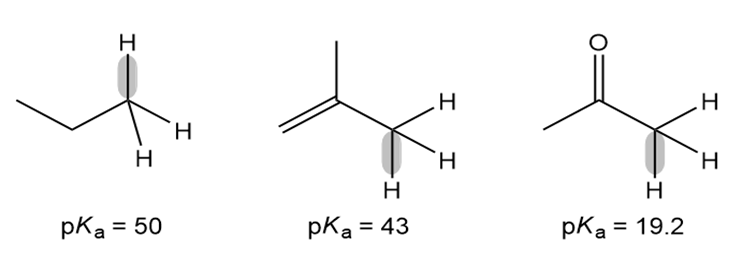
a. For each compound, draw the conjugate base, including all possible resonance structures.
b. Explain the observed trend in .
What do you think about this solution?
We value your feedback to improve our textbook solutions.