Chapter 2: Q. 2.41 (page 85)
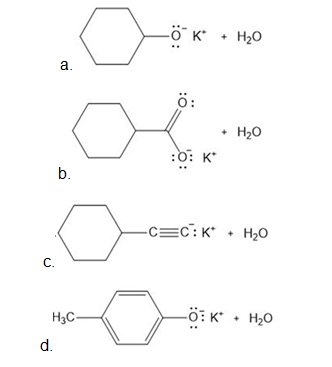
Draw the products formed from the acid–base reaction of KOH with each compound.
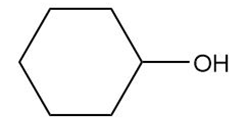
a.
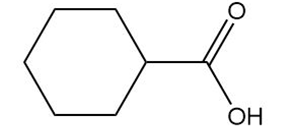
b.

c.
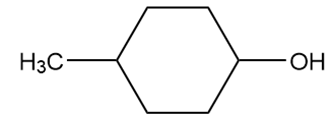
d.
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q. 2.41 (page 85)
Draw the products formed from the acid–base reaction of KOH with each compound.

a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
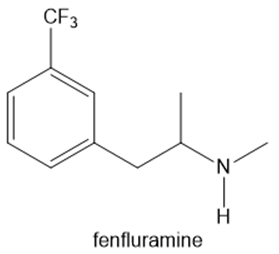
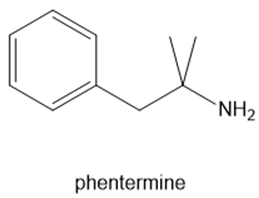
Fenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid ?
Which H atom in vitamin C (ascorbic acid) is most acidic?

Answer the following questions about esmolol, a drug used to treat high blood pressure sold under the trade name Brevibloc.
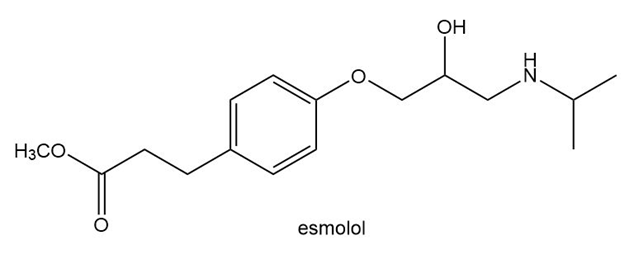
a. Label the most acidic hydrogen atom
b. What products are formed when esmolol is treated with NaH?
c. What products are formed when esmolol is treated with HCl?
d. Label all hybridized C atoms. in esmolol.
e. Label the only trigonal pyramidal atom.
f. Label all C’s that bear a charge.
Molecules like acetamide can be protonated on either their O or N atoms when treated with a strong acid like HCl. Which site is more readily protonated and why?
Classify each compound as a Lewis base, a Brønsted -Lowry base, both or neither.
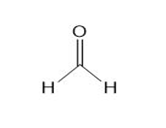
a.
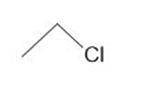
b.
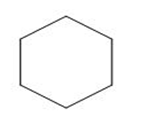
c.
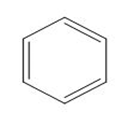
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.