Chapter 2: Q. 2.39 (page 85)
What is the conjugate base of each acid?
a.
b.
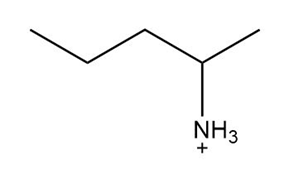
c.
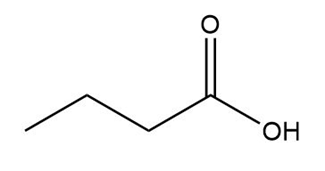
d.
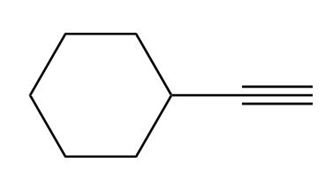
Short Answer
a.
b. role="math" localid="1649233356230"
c.
d.
Learning Materials
Features
Discover
Chapter 2: Q. 2.39 (page 85)
What is the conjugate base of each acid?
a.
b.

c.

d.

a.
b. role="math" localid="1649233356230"
c.
d.
All the tools & learning materials you need for study success - in one app.
Get started for free
Molecules like acetamide can be protonated on either their O or N atoms when treated with a strong acid like HCl. Which site is more readily protonated and why?
Atenolol is a (beta) blocker, a drug used to treat high blood pressure. Which of the indicated N-H bonds is more acidic? Explain your reasoning.
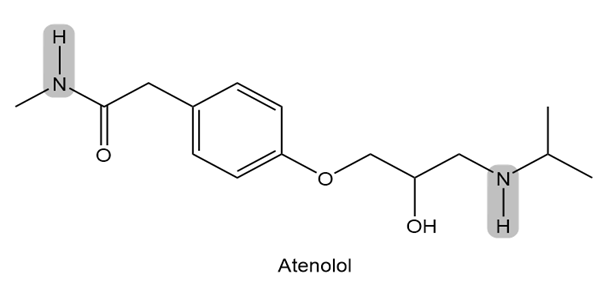
Which compound in each pair is the stronger acid?
a.
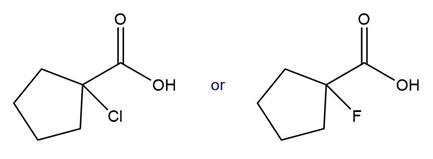
b.
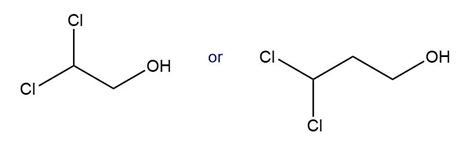
c.
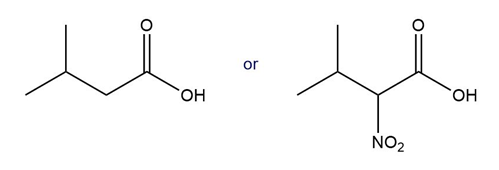
Label the three most acidic hydrogen atoms in lactic acid, , and rank them in order of decreasing acidity. Explain your reasoning.
Which species are Lewis bases?
a.
b.
c.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.