Chapter 2: 2.9 (page 68)
Rank the conjugate bases of each group of acids in order of increasing basicity:
a.
b.
Short Answer
a. The conjugate bases can be ranked as follows:
.
b. The conjugate base can be ranked as follows:
.
Learning Materials
Features
Discover
Chapter 2: 2.9 (page 68)
Rank the conjugate bases of each group of acids in order of increasing basicity:
a.
b.
a. The conjugate bases can be ranked as follows:
.
b. The conjugate base can be ranked as follows:
.
All the tools & learning materials you need for study success - in one app.
Get started for free
Atenolol is a (beta) blocker, a drug used to treat high blood pressure. Which of the indicated N-H bonds is more acidic? Explain your reasoning.
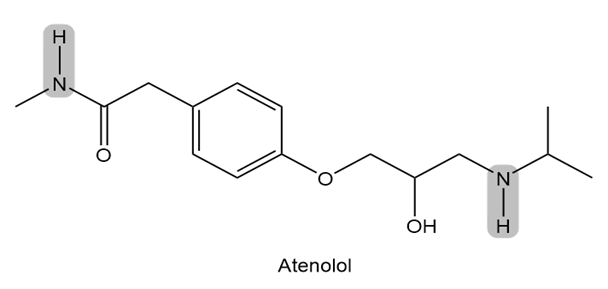
What is for each compound? Use a calculator when necessary.
a.
b.
c.
Dimethyl ether and ethanol are isomers, but has a role="math" localid="1649252571433" of 40 and has a of 16. Why are these values so different?
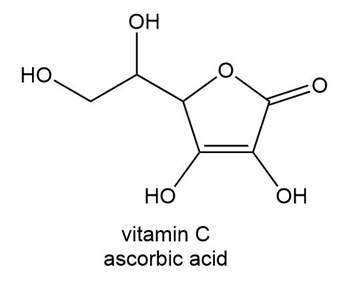
Which H atom in vitamin C (ascorbic acid) is most acidic?
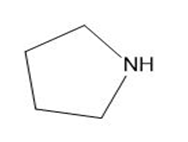
Draw the products formed from the acid-base reaction of HCl with each compound.
a.
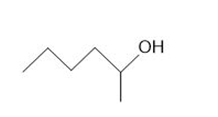
b.
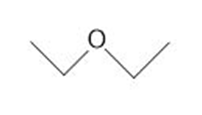
c.
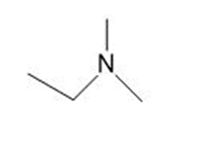
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.