Chapter 2: 2.26 (page 78)
Which proton in each of the following drugs is most acidic? THC is the active component in marijuana, and ketoprofen is an anti-inflammatory agent.
a.
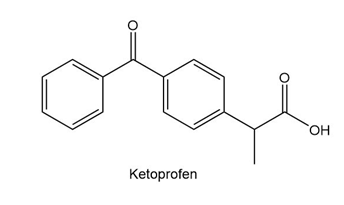
b.
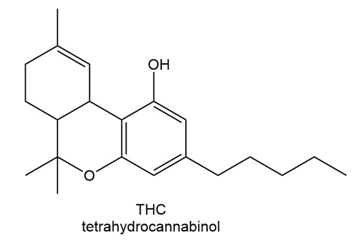
Short Answer
a.
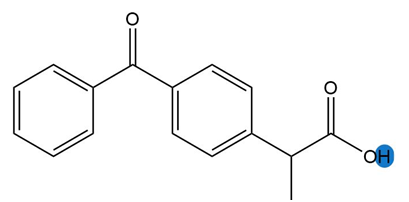
b.
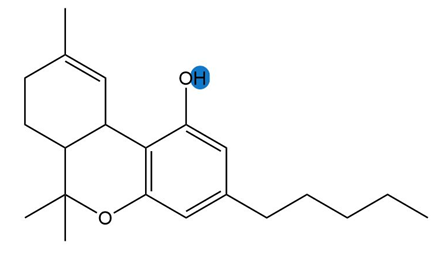
Learning Materials
Features
Discover
Chapter 2: 2.26 (page 78)
Which proton in each of the following drugs is most acidic? THC is the active component in marijuana, and ketoprofen is an anti-inflammatory agent.
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
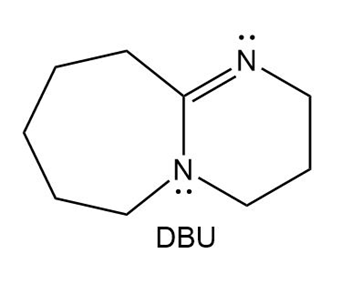
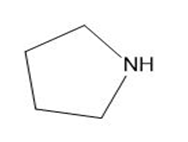
DBU, 1,8-diazabicyclo undec-7-ene, is a base we will encounter in elimination reactions in Chapter 8. Which N atom is more basic in DBU? Explain your choice.
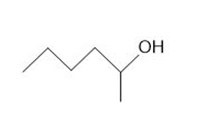
Draw the products formed from the acid-base reaction of HCl with each compound.
a.
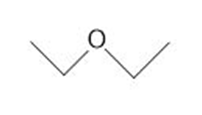
b.
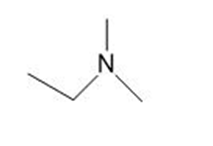
c.
d.
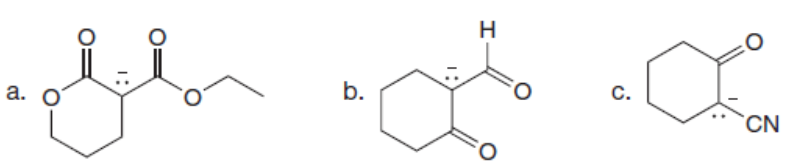
Question: Draw additional resonance structures for each anion.
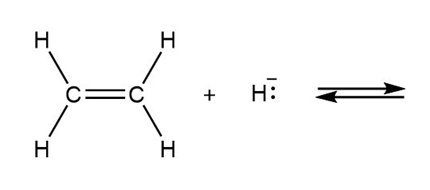
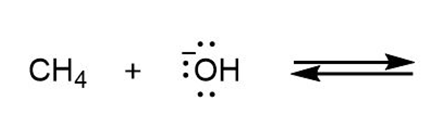
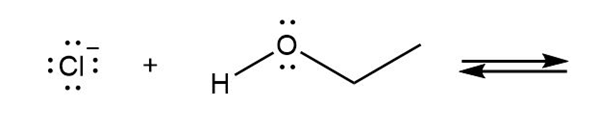
Draw the products of each reaction and determine the direction of equilibrium.
a.
b.
c.
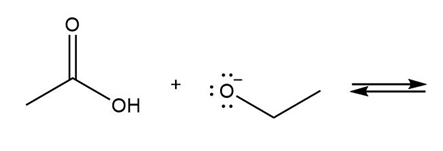
d.
Label the three most acidic hydrogen atoms in lactic acid, , and rank them in order of decreasing acidity. Explain your reasoning.
What do you think about this solution?
We value your feedback to improve our textbook solutions.