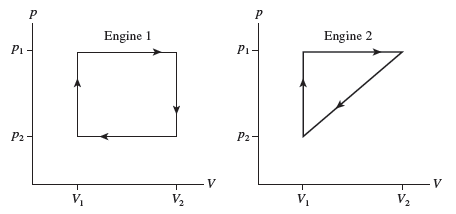
Chapter 21: Q. 6 (page 593)
shows the thermodynamic cycles of two heat engines. Which heat engine has the larger thermal efficiency? Or
are they the same? Explain.
Short Answer
The thermal efficiency
Learning Materials
Features
Discover
Chapter 21: Q. 6 (page 593)
shows the thermodynamic cycles of two heat engines. Which heat engine has the larger thermal efficiency? Or
are they the same? Explain.

The thermal efficiency
All the tools & learning materials you need for study success - in one app.
Get started for free
A Carnot heat engine operates between reservoirs at and . If the engine extracts of energy from the hot reservoir per cycle, how many cycles will it take to lift a mass a height of ?
The first and second laws of thermodynamics are sometimes stated as "You can't win" and "You can't even break even." Do these sayings accurately characterize the laws of thermodynamics as applied to heat engines? Why or why not?
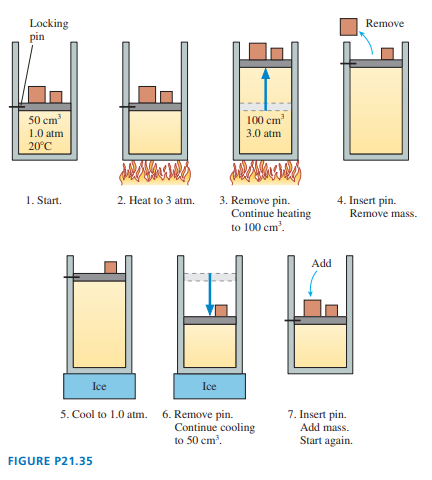
FIGURE P21.35 shows a heat engine going through one cycle. The gas is diatomic. The masses are such that when the pin is removed, in steps and , the piston does not move.
a. Draw the diagram for this heat engine.
b. How much work is done per cycle?
c. What is this engine’s thermal efficiency?
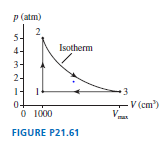
A heat engine using of helium as the working substance follows the cycle shown in FIGURE P.
a. Determine the pressure, temperature, and volume of the gas at points ,, and .
b. What is the engine's thermal efficiency?
c. What is the maximum possible efficiency of a heat engine that operates between and?
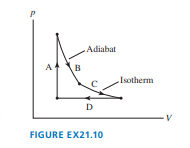
The cycle of FIGURE EX consists of four processes. Make a table with rows labeled to and columns labeled , , and . Fill each box in the table with ,, or to indicate whether the quantity increases, decreases, or stays the same during that process.
What do you think about this solution?
We value your feedback to improve our textbook solutions.