Chapter 18: A Macroscopic Description of Matter
Q.
A hollow aluminum sphere with outer diameter 10.0 cm has a
mass of 690 g. What is the sphere’s inner diameter?
Q. 1
Rank in order, from highest to lowest, the temperatures
T1 = 0 K, T2 = 0 C, and T3 = 0 F.
Q. 1
What volume of water has the same mass as 100 cm3 of gold?
Q. 10
What volume of aluminum has the same number of atoms as 10 cm3 of mercury?
Q. 10
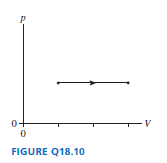
A gas under goes the process shown in FIGURE Q18.10. By what factor does the temperature change?
Q. 11
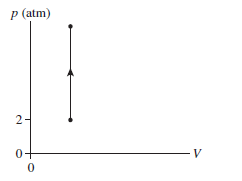
The temperature increases from 300 K to 1200 K as a gas undergoes
the process shown in FIGURE Q18.11. What is the final
pressure?
Q. 11
The lowest and highest natural temperatures ever recorded on earth are -129°F in Antarctica and 134°F in Death Valley. What are these temperatures in °C and in K?
Q. 12
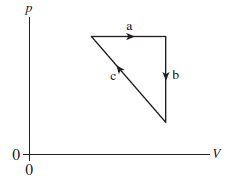
A student is asked to sketch a pV diagram for a gas that goes
through a cycle consisting of (a) an isobaric expansion, (b) a
constant-volume reduction in temperature, and (c) an isothermal
process that returns the gas to its initial state. The student draws
the diagram shown in FIGURE Q18.12. What, if anything, is wrong
with the student’s diagram?
Q. 12
At what temperature does the numerical value in °F match the numerical value in °C?
Q. 13
A demented scientist creates a new temperature scale, the “Z scale.” He decides to call the boiling point of nitrogen 0°Z and the melting point of iron 1000°Z.
a. What is the boiling point of water on the Z scale?
b. Convert 500°Z to degrees Celsius and to kelvins.
