Chapter 18: Q. 10 (page 511)
What volume of aluminum has the same number of atoms as 10 cm3 of mercury?
Short Answer
The Volume of aluminum is
Learning Materials
Features
Discover
Chapter 18: Q. 10 (page 511)
What volume of aluminum has the same number of atoms as 10 cm3 of mercury?
The Volume of aluminum is
All the tools & learning materials you need for study success - in one app.
Get started for free
A gas is in a sealed container. By what factor does the gas temperature change if:
a. The volume is doubled and the pressure is tripled?
b. The volume is halved and the pressure is tripled?
An aquanaut lives in an underwater apartment 100 m beneath the surface of the ocean. Compare the freezing and boiling points of water in the aquanaut’s apartment to their values at the surface. Are they higher, lower, or the same? Explain.
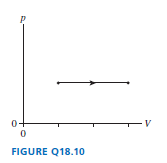
A gas under goes the process shown in FIGURE Q18.10. By what factor does the temperature change?
a. Is there the highest temperature at which ice can exist? If so what is it? If not, why not?
b. Is there a lowest temperature at which water vapour can exist?
if so, what is it? If not, why not?
A diving bell is a -tall cylinder closed at the upper end but open at the lower end. The temperature of the air in the bell is . The bell is lowered into the ocean until its lower end is deep. The temperature at that depth is .
a. How high does the water rise in the bell after enough time has passed for the air inside to reach thermal equilibrium?
b. A compressed-air hose from the surface is used to expel all the water from the bell. What minimum air pressure is needed to do this?
What do you think about this solution?
We value your feedback to improve our textbook solutions.