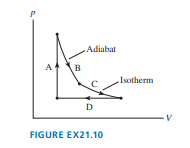
Chapter 21: Q. 10 (page 594)
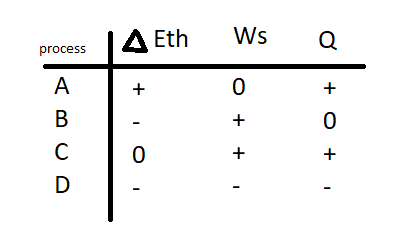
The cycle of FIGURE EX consists of four processes. Make a table with rows labeled to and columns labeled , , and . Fill each box in the table with ,, or to indicate whether the quantity increases, decreases, or stays the same during that process.
Short Answer
Result can be found taking into consideration that the processes are isochoric, adiabatic, isothermal and isobaric from A to D respectively.