Chapter 21: Q. 35 (page 596)
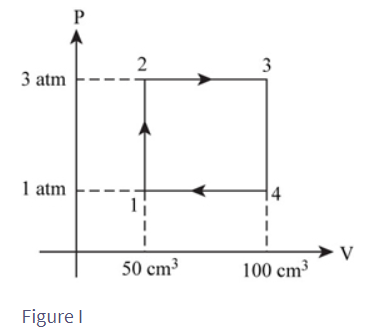
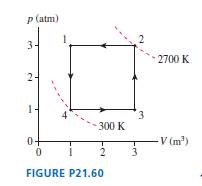
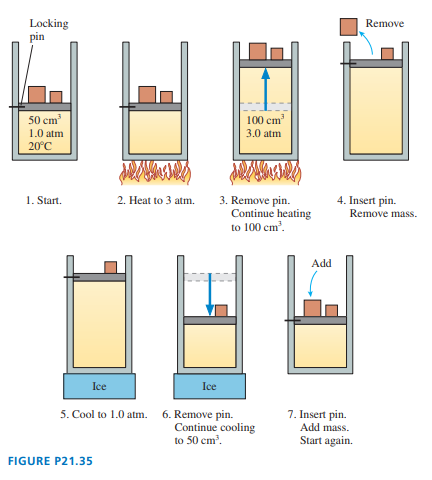
FIGURE P21.35 shows a heat engine going through one cycle. The gas is diatomic. The masses are such that when the pin is removed, in steps and , the piston does not move.
a. Draw the diagram for this heat engine.
b. How much work is done per cycle?
c. What is this engine’s thermal efficiency?
Short Answer
a.
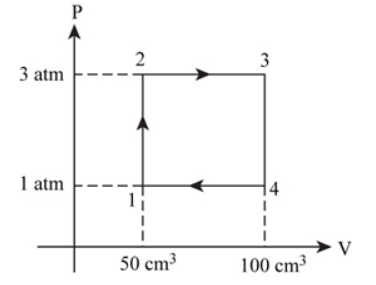
The diagram for heat engine is show above
b. The work done per cycle is
c. The thermal efficiency of heat engine is localid="1649578271077"