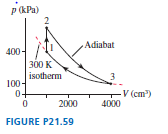
Chapter 21: Q. 59 (page 597)
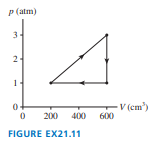
A heat engine uses a diatomic gas that follows the cycle in FIGURE P.
a. Determine the pressure, volume, and temperature at point
b. Determine , and
for each of the three processes. Put your results in a FIGURE Ptable for easy reading.
c. How much work does this engine do per cycle and what is its thermal efficiency?
Short Answer
a. Pressure,volume and temperature value is ,,.
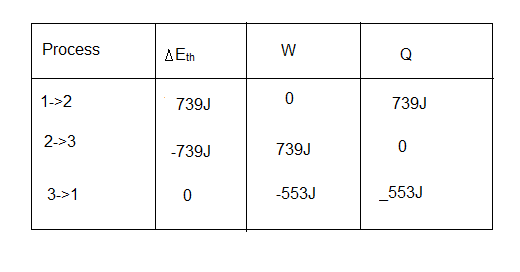
b. The table of result is  .
.
c. Work done for engine is ,thermal efficiency is.