Chapter 21: Q. 58 (page 597)
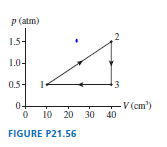
A heat engine using a monatomic gas follows the cycle shown in FIGURE P.
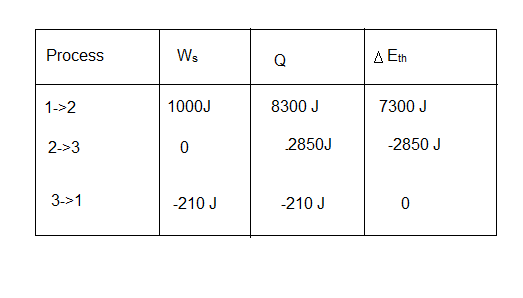
a. Find , and for each process in the cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
Short Answer
a. Table for process of cycle is  .
.
b. The thermal efficiency is.