The FIGURE shows the Diesel cycle. It is similar to the Otto cycle (see Problem), but there are two important differences. First, the fuel is not admitted until the air is fully compressed at point . Because of the high temperature at the end of adiabatic compression, the fuel begins to burn spontaneously. (There are no spark plugs in a diesel engine!) Second, combustion takes place more slowly, with fuel continuing to be injected. This makes the ignition stage a constant-pressure process. The cycle shown, for one cylinder of a diesel engine, has a displacement localid="1650278379201" of localid="1650278390412" and a compression ratio localid="1650278397535" These are typical values for a diesel truck. The engine operates with intake air localid="1650278425123" at localid="1650278430754" and localid="1650278435051" pressure. The quantity of fuel injected into the cylinder has a heat of combustion of localid="1650278440305" .
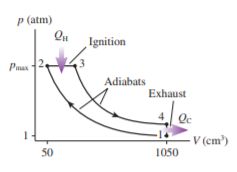
. Find , , and at each of the four corners of the cycle. Display your results in a table.
. What is the network done by the cylinder during one full cycle?
. What is the thermal efficiency of this engine?
. What is the power output in kW and horsepower of an eight-cylinder diesel engine running at ?



