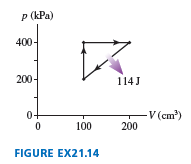
Chapter 21: Q. 60 (page 598)
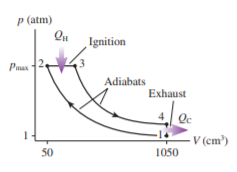
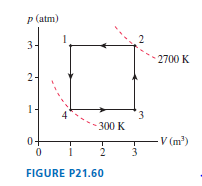
FIGURE Pis the diagram of Example , but now the device is operated in reverse.
a. During which processes is heat transferred into the gas?
b. Is this, heat extracted from a hot reservoir, or , heat extracted from a cold reservoir? Explain.
c. Determine the values ofand.
Hint: The calculations have been done in Example and do not need to be repeated. Instead, you need to determine which processes now contribute to and which to .
d. Is the area inside the curve or ? What is its value?
e. The device is now being operated in a ccw cycle. Is it a refrigerator? Explain.
Short Answer
a. In process heat transferred into the gas.
b. Heat extracted from cold reservoir.
c. Heat of hot reservoir is ,cold reservoir is .
d. Area inside the curve is and it is counter clockwise.
e. Device is operated in a ccw cycle is not refrigerator.