Chapter 21: Q. 14 (page 594)
What are (a) andand (b) the thermal efficiency for the heat engine shown in
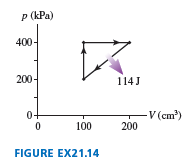
Short Answer
a.The work done by the gas will be numerically equal to the area under the graph is,is.
b.The efficiency for a heat engine is.
Learning Materials
Features
Discover
Chapter 21: Q. 14 (page 594)
What are (a) andand (b) the thermal efficiency for the heat engine shown in

a.The work done by the gas will be numerically equal to the area under the graph is,is.
b.The efficiency for a heat engine is.
All the tools & learning materials you need for study success - in one app.
Get started for free
The engine that powers a crane burns fuel at a flame temperature of 2000°C. It is cooled by 20°C air. The crane lifts a 2000 kg steel girder 30 m upward. How much heat energy is transferred to the engine by burning fuel if the engine is 40% as efficient as a Carnot engine?
A heat engine with of the Carnot efficiency operates between reservoirs at and . The engine inputs heat energy at an average rate of while compressing a spring in . What is the spring constant?
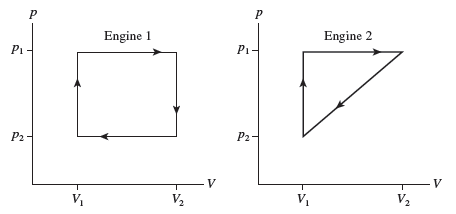
shows the thermodynamic cycles of two heat engines. Which heat engine has the larger thermal efficiency? Or
are they the same? Explain.
A Carnot refrigerator operates between reservoirs at and in a room. The refrigerator is a box. Five of the walls are perfect insulators, but the sixth is a thick piece of stainless steel. What electric power does the refrigerator require to maintain the inside temperature at ?
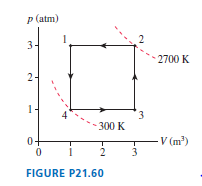
FIGURE Pis the diagram of Example , but now the device is operated in reverse.
a. During which processes is heat transferred into the gas?
b. Is this, heat extracted from a hot reservoir, or , heat extracted from a cold reservoir? Explain.
c. Determine the values ofand.
Hint: The calculations have been done in Example and do not need to be repeated. Instead, you need to determine which processes now contribute to and which to .
d. Is the area inside the curve or ? What is its value?
e. The device is now being operated in a ccw cycle. Is it a refrigerator? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.