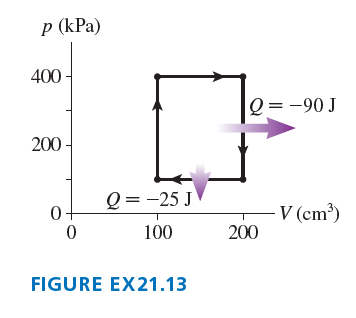
Chapter 21: Q. 13 (page 594)
What are and and the thermal efficiency for the heat engine shown in?
Short Answer
The values are
The thermal efficiency for the heat engine is
Learning Materials
Features
Discover
Chapter 21: Q. 13 (page 594)
What are and and the thermal efficiency for the heat engine shown in?

The values are
The thermal efficiency for the heat engine is
All the tools & learning materials you need for study success - in one app.
Get started for free
A nuclear power plant generates of heat energy from nuclear reactions in the reactor’s core. This energy is used to boil water and produce high-pressure steam at . The steam spins a turbine, which produces of electric power, then the steam is condensed and the water is cooled to before starting the cycle again.
a. What is the maximum possible thermal efficiency of the power plant?
b. What is the plant’s actual efficiency?
c. Cooling water from a river flows through the condenser (the low-temperature heat exchanger) at the rate of ( million gallons per hour). If the river water enters the condenser at , what is its exit temperature?
A heat engine extracts of heat from the hot reservoir each cycle and exhausts of heat. What are (a) the thermal efficiency and (b) the work done per cycle?
A Carnot refrigerator operating between andextracts heat from the cold reservoir at the rate. What are (a) the coefficient of performance of this refrigerator, (b) the rate at which work is done on the refrigerator, and (c) the rate at which heat is exhausted to the hot side?
What are (a) the heat extracted from the cold reservoir and (b) the coefficient of performance for the refrigerator shown in Figure Ex-?
A heat engine operating between energy reservoirs atand has of the maximum possible efficiency. How much energy must this engine extract from the hot reservoir to do of work?
What do you think about this solution?
We value your feedback to improve our textbook solutions.