Chapter 13: Q13-40E (page 419)
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, or diastereotopic:
a.
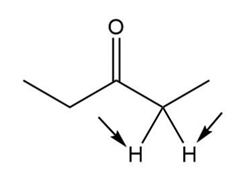
b.
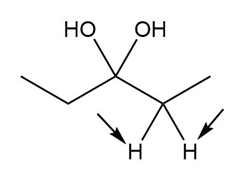
c.
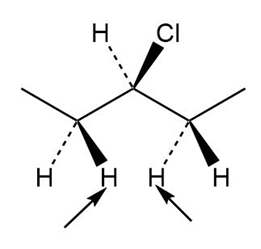
Short Answer
Enantiotopic
Diastereotopic
Diastereotopic
Learning Materials
Features
Discover
Chapter 13: Q13-40E (page 419)
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, or diastereotopic:
a.

b.

c.

Enantiotopic
Diastereotopic
Diastereotopic
All the tools & learning materials you need for study success - in one app.
Get started for free
Classify the resonances in the13C NMR spectrum of methyl propanoate,.
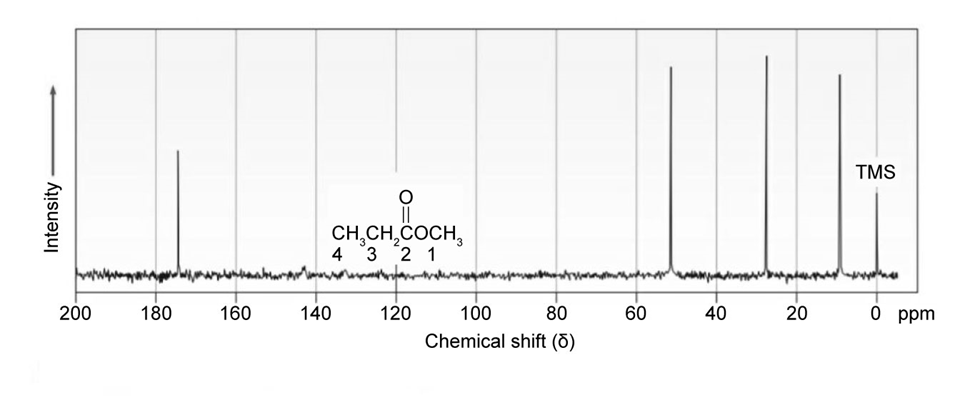
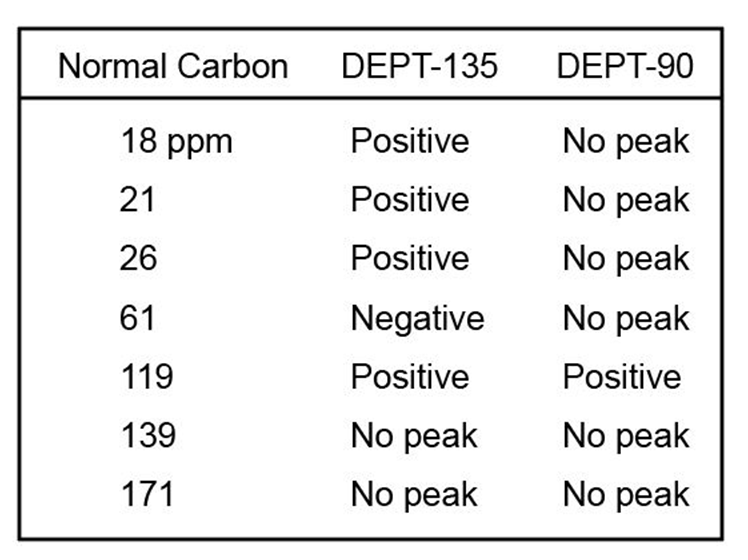
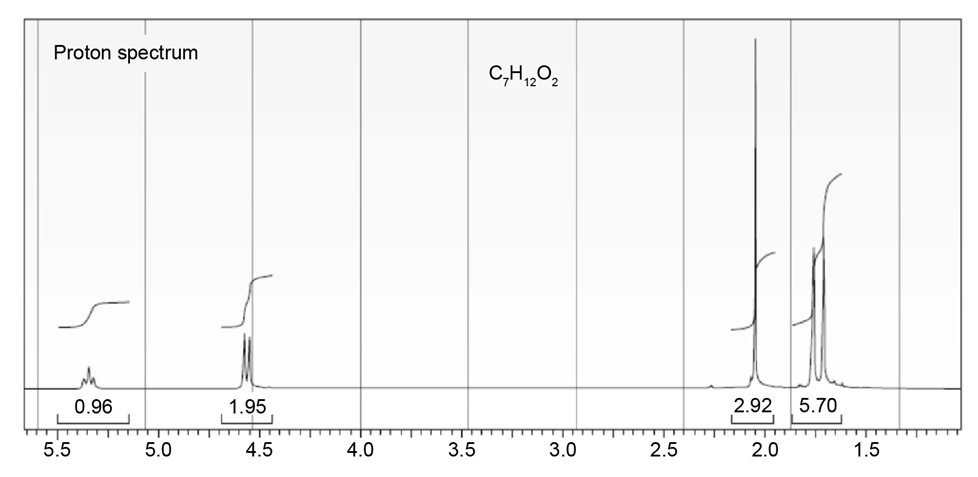
The proton NMR spectrum of a compound with the formula C7H12O2is
shown. The infrared spectrum displays a strong band at 1738 cm-1 and
a weak band at 1689 cm-1 . The normal carbon-13 and the DEPT experimental
results are tabulated. Draw the structure of this compound.
Suppose you ran a DEPT-135 spectrum for each substance in Problem
13-47. Which carbon atoms in each molecule would show positive
peaks, and which would show negative peaks?
The following NMR peaks were recorded on a spectrometer operating at
200 MHz. Convert each into O1units.
(a) CH3CI; 1454Hz
(b) CHCI3; 610Hz
(c) CH3OH; 693Hz
(d) CH2CI2; 1060Hz
Propose a structure for compound E, , which has the following NMR spectral data:
Compound E Broadband-decoupled NMR: 19.1, 28.0, 70.5, 129.0, 129.8, 165.8 DEPT-90: 28.0, 129.8 DEPT-135: positive peaks at 19.1, 28.0, 129.8 ; negative peaks at 70.5, 129.0
What do you think about this solution?
We value your feedback to improve our textbook solutions.