Chapter 13: Q13-19P (page 413)
Classify the resonances in the13C NMR spectrum of methyl propanoate,.
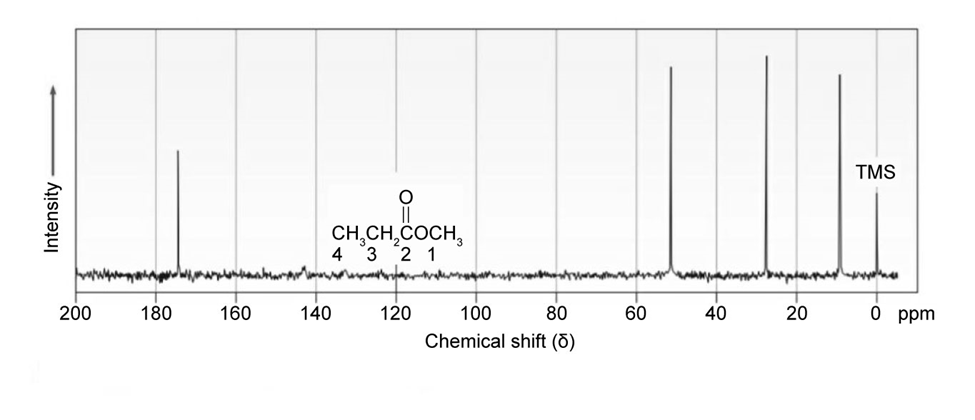
Short Answer
 NMR Spectra
NMR Spectra
Learning Materials
Features
Discover
Chapter 13: Q13-19P (page 413)
Classify the resonances in the13C NMR spectrum of methyl propanoate,.

 NMR Spectra
NMR Spectra
All the tools & learning materials you need for study success - in one app.
Get started for free
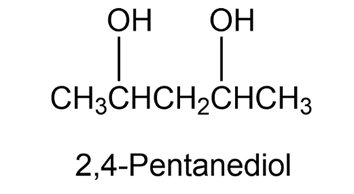
A NMR spectrum of commercially available 2,4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8, and 68.1 d. Explain.
We saw in Section 9-3 that addition of H-Br to a terminal alkyne leads to the Markovnikov addition product, with the Br bonding to the more highly substituted carbon. How could you use 13C NMR to identify the product of theaddition of 1 equivalent of H-Br to 1-hexyne?
Compound F, a hydrocarbon with = 96 in its mass spectrum, undergoes reaction with HBr to yield compound G. Propose structures for F and G, whose NMR spectral data are given below.
a. Compound F Broadband-decoupled NMR: 27.6, 29.3, 32.2, 132.4 d DEPT-90: 132.4 d DEPT-135: positive peak at 132.4 d; negative peaks at 27.6, 29.3, 32.2 d
b. Compound G Broadband-decoupled NMR: 25.1, 27.7, 39.9, 56.0 d DEPT-90: 56.0 d DEPT-135: positive peak at 56.0 d; negative peaks at 25.1, 27.7, 39.9 d
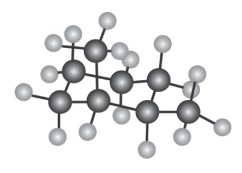
Question: How many electronically non-equivalent kinds of protons and how
many kinds of carbons are present in the following compound? Don’t
forget that cyclohexane rings can ring-flip.
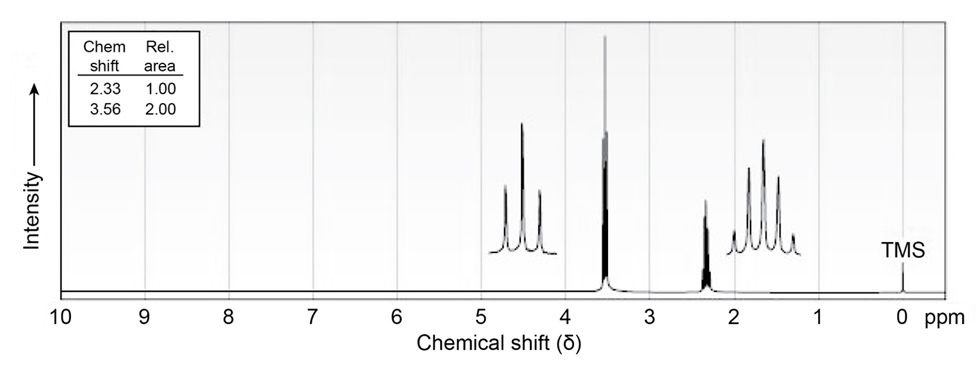
The compound whose 1H NMR spectrum is shown has the molecular
formula C3H6Br2. Propose a structure.
What do you think about this solution?
We value your feedback to improve our textbook solutions.