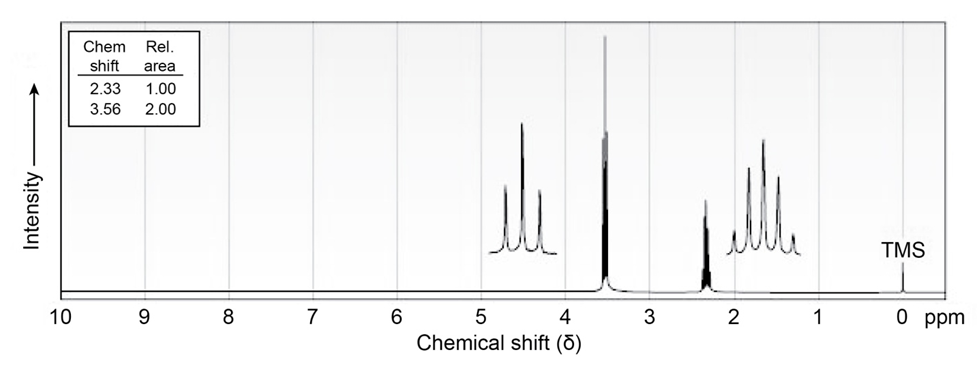
Chapter 13: Q13.53E (page 419)
The compound whose 1H NMR spectrum is shown has the molecular
formula C3H6Br2. Propose a structure.
Short Answer
Learning Materials
Features
Discover
Chapter 13: Q13.53E (page 419)
The compound whose 1H NMR spectrum is shown has the molecular
formula C3H6Br2. Propose a structure.


All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the splitting pattern for each kind of hydrogen in the following molecules:
(a)
(b)
(c) trans-2-Butene
Assign a chemical shift to each carbon in 6-methyl-5-hepten-2-ol.
Propose structures for compounds that fit the following 1H NMR data:
a.
2.18(3H, singlet)
4.16(2H, doublet j=7Hz)
5.71(1H, triplet j=7Hz)
b.
1.30(9H, singlet)
7.30(5H, singlet)
c.
2.11(3H, singlet)
3.52(2H, triplet j=6Hz)
5.71(2H, triplet j=6Hz)
d.
2.15(2H, quintent j=7Hz)
2.75(2H, triplet j=7Hz)
3.38(2H, triplet j=7Hz)
7.22(5H, singlet)
Question: 3-Bromo-1-phenyl-1-propene shows a complex NMR spectrum in which the vinylic proton at C2 is coupled with both the C1 vinylic proton (J = 16 Hz) and the C3 methylene protons (J = 8 Hz). Draw a tree diagram for the C2 proton signal, and account for the fact that a five-line multiplet is observed .
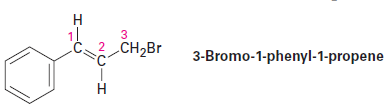
Propose a structure for compound E, , which has the following NMR spectral data:
Compound E Broadband-decoupled NMR: 19.1, 28.0, 70.5, 129.0, 129.8, 165.8 DEPT-90: 28.0, 129.8 DEPT-135: positive peaks at 19.1, 28.0, 129.8 ; negative peaks at 70.5, 129.0
What do you think about this solution?
We value your feedback to improve our textbook solutions.