Chapter 13: Q13-27E (page 419)
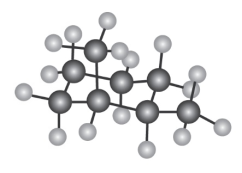
Question: How many electronically non-equivalent kinds of protons and how
many kinds of carbons are present in the following compound? Don’t
forget that cyclohexane rings can ring-flip.
Short Answer
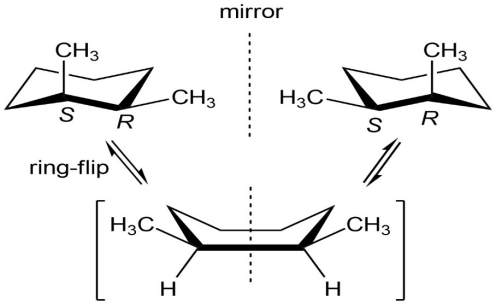
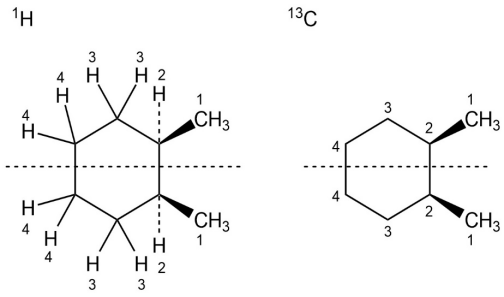
If you assign R, S configuration to the carbons bonded to methyl group, it is apparent that cis-1,2-dimethylcyclohexane is a mesocompound. When the cyclohexane ring undergoes a ring-flip, the ring passes through an intermediate that has a plane of symmetry. Both the13C NMR spectrum and the1H NMR spectrum show 4 peaks.