Chapter 13: Q13.44E (page 419)
Propose structures for compounds that fit the following NMR data:
(a) C5H10O
0.95(6H,double, J = 7Hz)
2.10(3H,singlet)
2.43(1H, mutiplet)
(b) C3H5Br
2.32(3H,singlet)
5.35 (3H,singlet)
5.45(1H, broad signet )
Learning Materials
Features
Discover
Chapter 13: Q13.44E (page 419)
Propose structures for compounds that fit the following NMR data:
(a) C5H10O
0.95(6H,double, J = 7Hz)
2.10(3H,singlet)
2.43(1H, mutiplet)
(b) C3H5Br
2.32(3H,singlet)
5.35 (3H,singlet)
5.45(1H, broad signet )
All the tools & learning materials you need for study success - in one app.
Get started for free
Carboxylic acids react with alcohols (R'OH) in the presence of an acid catalyst. The reaction product of propanoic acid with methanol has the following spectroscopic properties. Propose a structure.
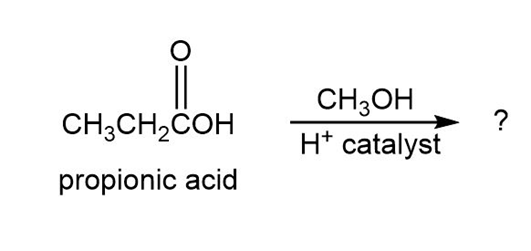
MS: = 88
IR: 1735
NMR: 1.11 (3 H, triplet, J 5 7 Hz); 2.32 (2 H, quartet, J 5 7 Hz); 3.65 (3 H, singlet) NMR: 9.3, 27.6, 51.4, 174.6
Predict the splitting pattern for each kind of hydrogen in the following molecules:
(a)
(b)
(c) trans-2-Butene
2-Chloropropene shows signals for three kinds of protons in its 1H NMR spectrum. Explain.
Propose a structure for compound C, which has = 86 in its mass spectrum, an IR absorption at 3400, and the following NMR spectral data:
Compound C Broadband-decoupled NMR: 30.2, 31.9, 61.8, 114.7, 138.4 d DEPT-90: 138.4 d DEPT-135: positive peak at 138.4 d; negative peaks at 30.2, 31.9, 61.8, 114.7 d
Question: When measured on a spectrometer operating at 200 MHz, chloroform shows a single sharp absorption at 7.3.
(a)How many parts per million downfield from TMS does chloroform absorb?
(b)How many hertz downfield from TMS would chloroform absorb if the measurement were carried out on a spectrometer operating at 360 MHz?
(c)What would be the position of the chloroform absorption inunits when measured on a 360 MHz spectrometer?
What do you think about this solution?
We value your feedback to improve our textbook solutions.