Chapter 13: Q13.45E (page 419)
Propose structures for the two compounds whose1H NMR spectra are
shown.
(a) C4H9Br
(b) C4H8CI2
Learning Materials
Features
Discover
Chapter 13: Q13.45E (page 419)
Propose structures for the two compounds whose1H NMR spectra are
shown.
(a) C4H9Br
(b) C4H8CI2
All the tools & learning materials you need for study success - in one app.
Get started for free
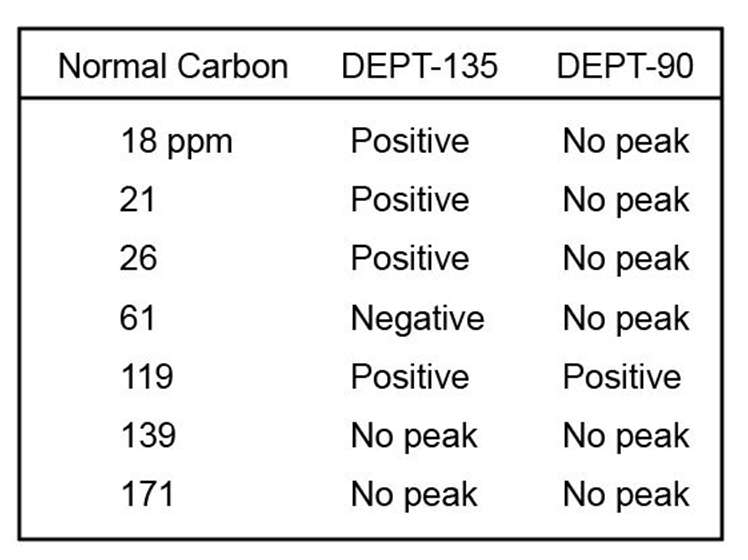
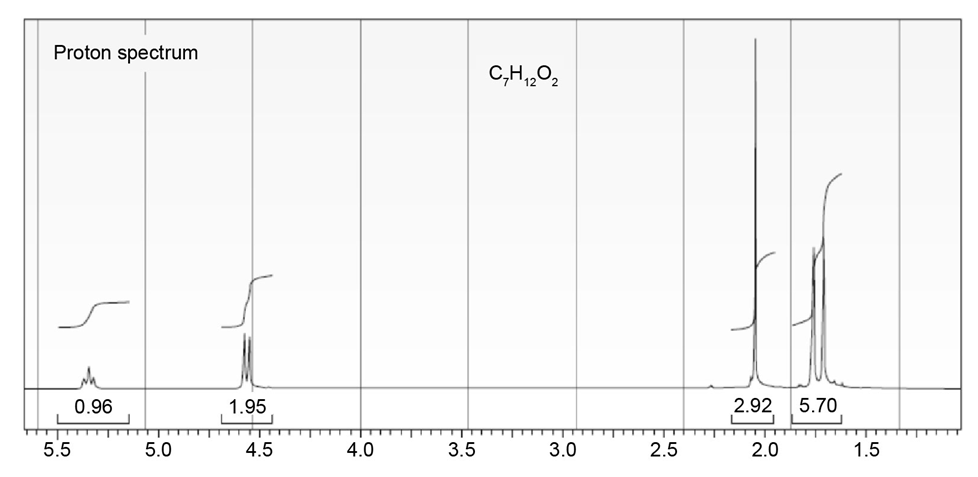
The proton NMR spectrum of a compound with the formula C7H12O2is
shown. The infrared spectrum displays a strong band at 1738 cm-1 and
a weak band at 1689 cm-1 . The normal carbon-13 and the DEPT experimental
results are tabulated. Draw the structure of this compound.
Propose a structure for compound C, which has = 86 in its mass spectrum, an IR absorption at 3400, and the following NMR spectral data:
Compound C Broadband-decoupled NMR: 30.2, 31.9, 61.8, 114.7, 138.4 d DEPT-90: 138.4 d DEPT-135: positive peak at 138.4 d; negative peaks at 30.2, 31.9, 61.8, 114.7 d
How many kinds of electronically non-equivalent protons are present in each of the following compounds, and thus how many NMR absorptions might you expect in each?
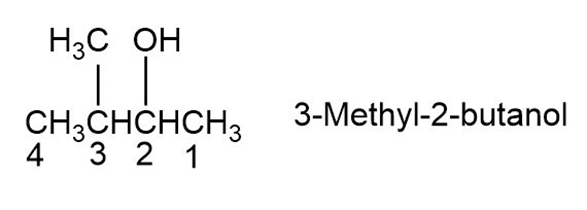
3-Methyl-2-butanol has five signals in its NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 d. Why are the two methyl groups attached to C3 non- equivalent? Making a molecular model should be helpful.
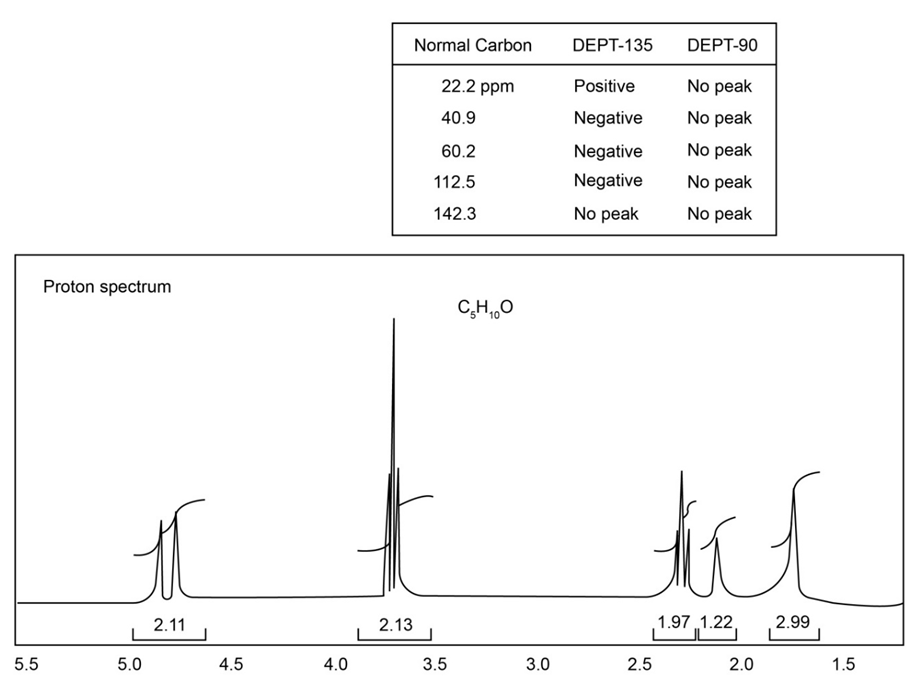
The proton NMR spectrum of a compound with the formula C5H10O isshown. The normal carbon-13 and the DEPT experimental results aretabulated. The infrared spectrum shows a broad peak at about3340 cm-1 and a medium-sized peak at about 1651 cm-1. Draw the structure of thiscompound.
What do you think about this solution?
We value your feedback to improve our textbook solutions.