Chapter 13: Q13-41E (page 419)
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, or diastereotopic:
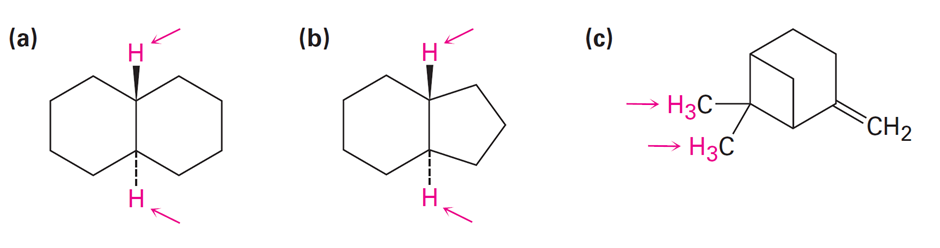
Short Answer
- Homotopic
- Enantiotopic
- Diastereotopic
Learning Materials
Features
Discover
Chapter 13: Q13-41E (page 419)
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, or diastereotopic:

All the tools & learning materials you need for study success - in one app.
Get started for free
Long-range coupling between protons more than two carbon atoms apart is sometimes observed when p bonds intervene. An example is found in 1-methoxy-1-buten-3-yne. Not only does the acetylenic proton, , couple with the vinylic proton , it also couples with the vinylic proton , four carbon atoms away. The data are:
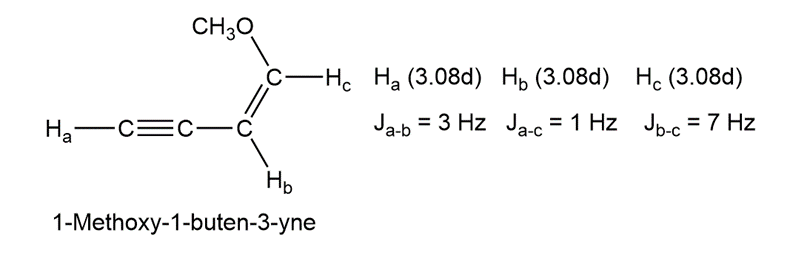
Construct tree diagrams that account for the observed splitting patterns of ,, and .
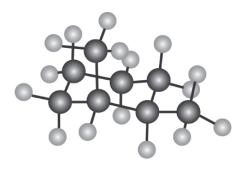
Question: How many electronically non-equivalent kinds of protons and how
many kinds of carbons are present in the following compound? Don’t
forget that cyclohexane rings can ring-flip.
Propose structures for compounds that fit the following descriptions:
(a) A hydrocarbon with seven lines in its 13C NMR spectrum
(b) A six-carbon compound with only five lines in its 13C NMR spectrum
(c) A four-carbon compound with three lines in its 13C NMR spectrum
Suppose you ran a DEPT-135 spectrum for each substance in Problem
13-47. Which carbon atoms in each molecule would show positive
peaks, and which would show negative peaks?
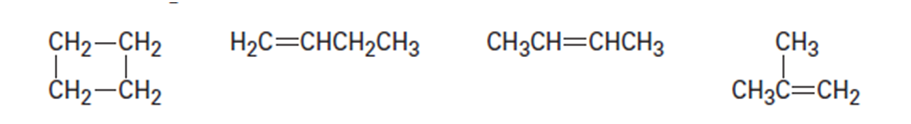
How could you use and1H 13C NMR to help distinguish the following
isomeric compounds of the formula C4H8?
What do you think about this solution?
We value your feedback to improve our textbook solutions.