Chapter 12: Q65E (page 354)
Question: 21-65 Propose a structure for a compound, , that has the following
IR and1H NMR spectra:
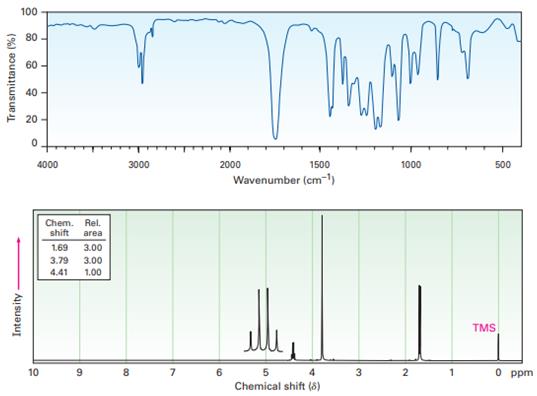
Short Answer
The structure for a compound
Learning Materials
Features
Discover
Chapter 12: Q65E (page 354)
Question: 21-65 Propose a structure for a compound, , that has the following
IR and1H NMR spectra:

The structure for a compound
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Propose structures for compounds that fit the following mass-spectral data:
(a) A hydrocarbon with
(b) A hydrocarbon with
(c) A hydrocarbon with
Question: 2-Methylpentane (C6H14)has the mass spectrum shown. Which peak
represents? Which is the base peak? Propose structures for fragment
ions of m/z=71, 57, 43, and 29. Why does the base peak have the mass it does?
Assume that you are carrying out the base-induced dehydrobromination of 3-Bromo-3-methyl pentane (Section 11-7) to yield an alkene. How could you use IR spectroscopy to tell which of three possible elimination products is formed, if one includes E/Z isomers?
Predict the product and provide the complete electron-pushing mechanism for the following two-step synthetic processes:
Carvone is an unsaturated ketone responsible for the odour of spearmint. If carvone hasin its mass spectrum and contains three double bonds and one ring, what is its molecular formula?
What do you think about this solution?
We value your feedback to improve our textbook solutions.