Chapter 12: Q24E (page 354)
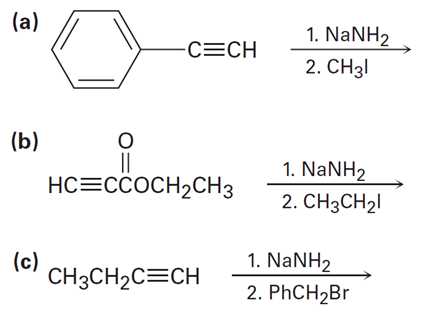
Predict the product and provide the complete electron-pushing mechanism for the following two-step synthetic processes:
Short Answer
(a)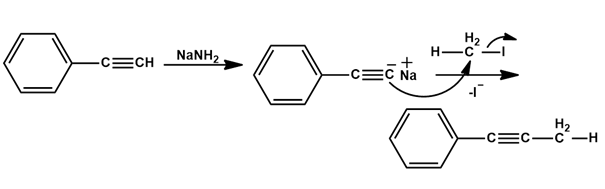
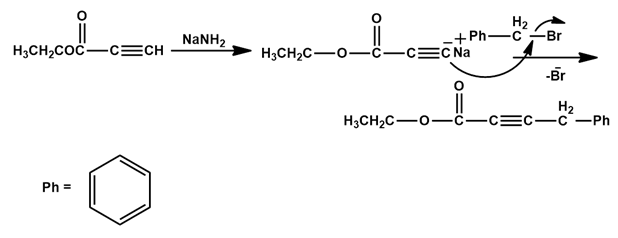
(b)
(c)
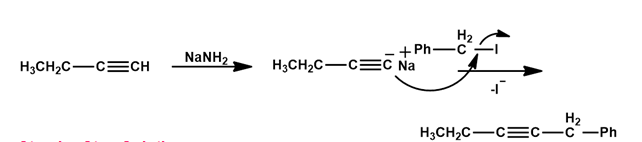
Learning Materials
Features
Discover
Chapter 12: Q24E (page 354)
Predict the product and provide the complete electron-pushing mechanism for the following two-step synthetic processes:

(a)
(b)

(c)
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What functional groups might the following molecules contain?
(a) A compound with a strong absorption at 1710
(b) A compound with a strong absorption at 1540
(c) A compound with strong absorptions at 1720 and 2500 to 3100
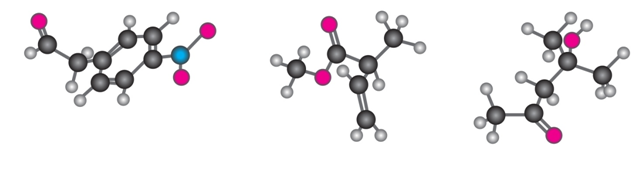
Where might the following compound have IR absorptions?
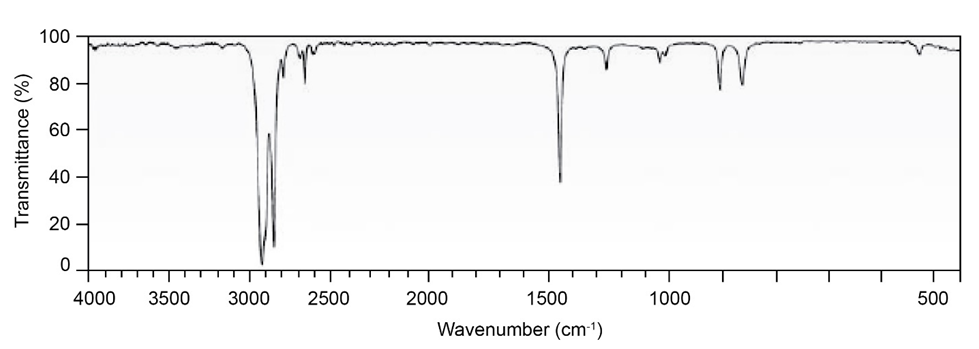
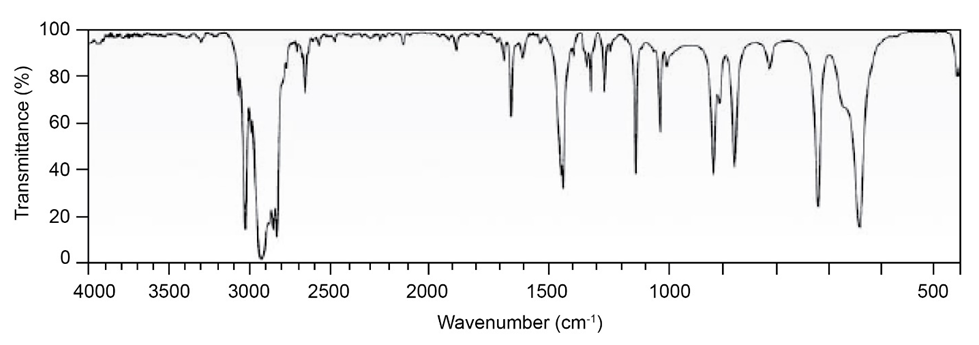
Question: Two infrared spectra are shown. One is the spectrum of cyclohexane,
and the other is the spectrum of cyclohexene. Identify them, and
explain your answer.
(a)
(b)
Assume that you are carrying out the dehydration of 1-methyl cyclohexanol to yield 1-methyl cyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete?
Question: By knowing the natural abundances of minor isotopes, it’s possible to
calculate the relative heights of and M+1 peaks. If 13C has a natural
abundance of 1.10%, what are the relative heights of the and
M+1 peaks in the mass spectrum of benzene, C6H6?
What do you think about this solution?
We value your feedback to improve our textbook solutions.