Chapter 12: Q12-12-12P (page 385)
Where might the following compound have IR absorptions?
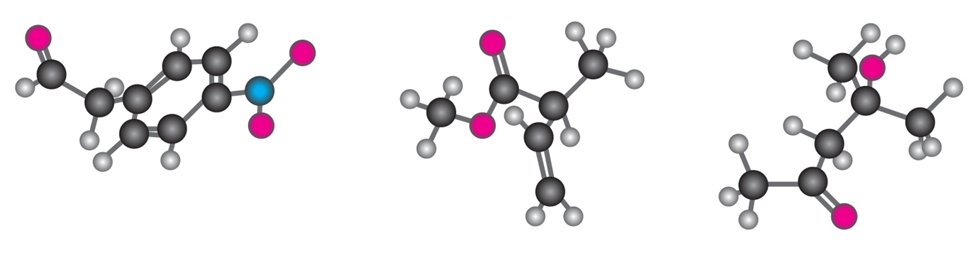
Short Answer
Absorption bands seen at 2210-2260 \(c{m^{ - 1}}\), 1690 \(c{m^{ - 1}}\) and 1640-1680 \(c{m^{ - 1}}\).
Learning Materials
Features
Discover
Chapter 12: Q12-12-12P (page 385)
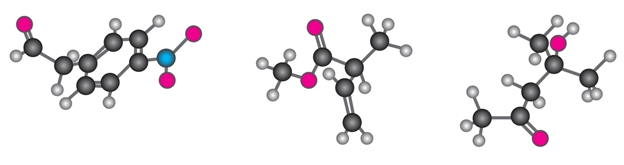
Where might the following compound have IR absorptions?

Absorption bands seen at 2210-2260 \(c{m^{ - 1}}\), 1690 \(c{m^{ - 1}}\) and 1640-1680 \(c{m^{ - 1}}\).
All the tools & learning materials you need for study success - in one app.
Get started for free
Where might the following compounds have IR absorptions?
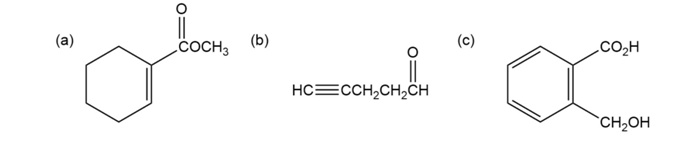
Question: At what approximate positions might the following compounds show
IR absorptions?
(b)
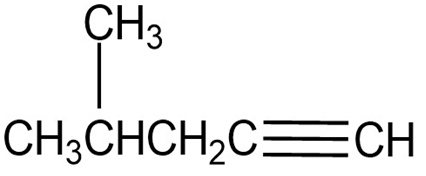
Question: Where might the following compound have IR absorptions?
Write molecular formulas for compounds that show the following molecularions in their high-resolution mass spectra, assuming that C, H, N,and O might be present. The exact atomic masses are: 1.00783(1H), 12.00000(12C) , 14.00307 (14N) , 15.99491(16O) .
(a) M+ = 98.0844
(b) M+= 123.0320
Question:The mass spectrum (a) and the infrared spectrum (b) of an unknown hydrocarbon are shown. Propose as many structures as you can.
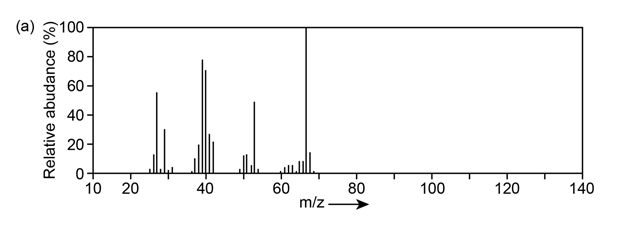
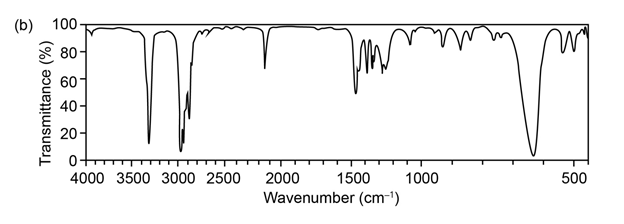
What do you think about this solution?
We value your feedback to improve our textbook solutions.