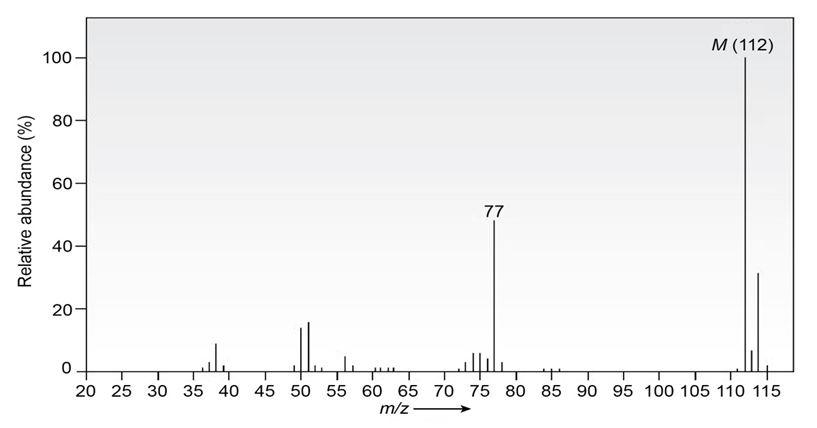
Chapter 12: Q-12-12-50E (page 385)
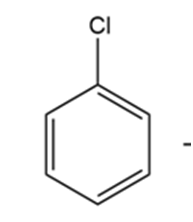
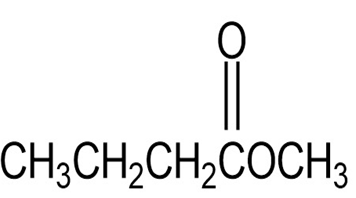
Question: The infrared spectrum of the compound with the mass spectrum shown below has strong absorbances at 1584, 1478, and . Propose a
structure consistent with the data.
Short Answer
Learning Materials
Features
Discover
Chapter 12: Q-12-12-50E (page 385)
Question: The infrared spectrum of the compound with the mass spectrum shown below has strong absorbances at 1584, 1478, and . Propose a
structure consistent with the data.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: By knowing the natural abundances of minor isotopes, it’s possible to
calculate the relative heights of and M+1 peaks. If 13C has a natural
abundance of 1.10%, what are the relative heights of the and
M+1 peaks in the mass spectrum of benzene, C6H6?
Propose structures for compounds that fit the following mass-spectraldata:
(a) A hydrocarbon with M+= 132
(b) A hydrocarbon with M+=166
(c) A hydrocarbon with M+= 84
At what approximate positions might the following compounds showIR absorptions?
(d)
Question: Propose structures for compounds that meet the following descriptions:
A non-optically active compound with an IR absorption at.
In light of the nitrogen rule mentioned in Problem 12-17, what is themolecular formula of pyridine, M+=79?
What do you think about this solution?
We value your feedback to improve our textbook solutions.