Chapter 12: Structure Determination: Mass Spectroscopy and Infrared Spectroscopy
Q13P
Beginning with acetylene and any alkyl halide needed, how would you synthesize the following compounds?
(a)Decane
(b)2,2-Dimethylhexane
(c)Hexanal
(d)2-Heptanone
Q13P
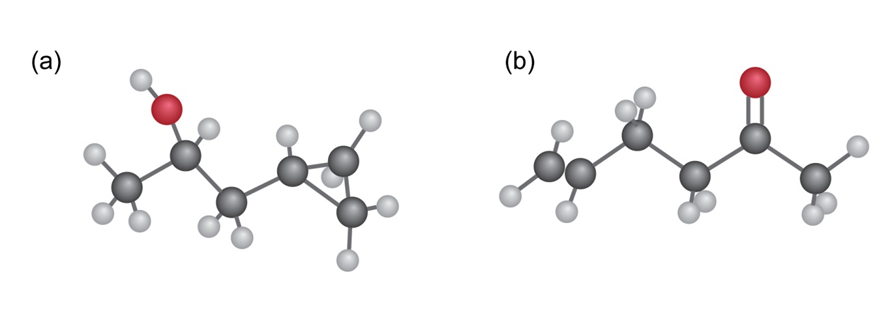
Show how you could prepare the following compounds from 4-methyl-3- penten-2-one,
(a)
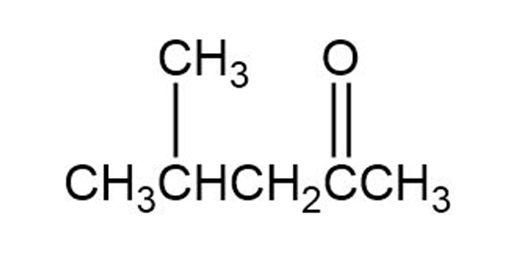
(b)
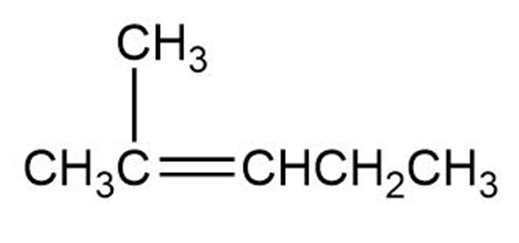
(c)
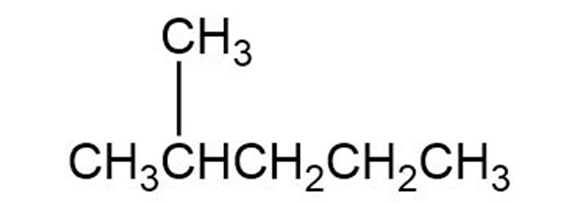
Q 13 P
Question: Show the structures of the fragments you would expect in the mass spectra of the following molecules:
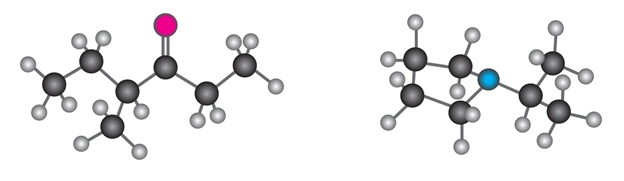
Q14E
Name the following alkynes, and predict the products of their reaction
With:
(1) in the presence of a Lindlar’s catalyst
(2) in the presence of
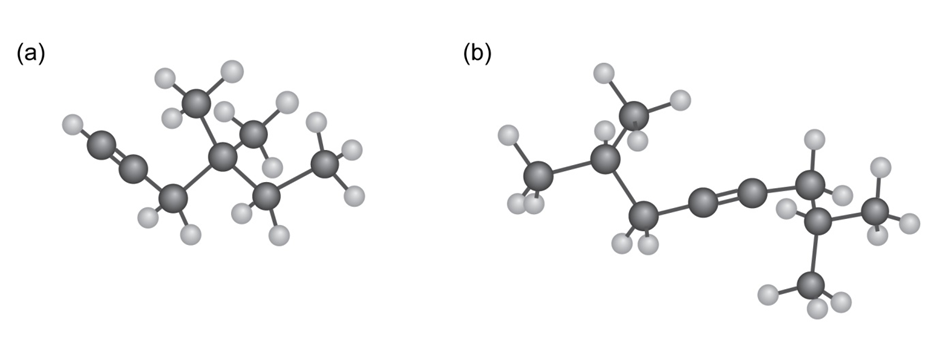
Q14P
Propose structures for compounds that fit the following mass-spectraldata:
(a) A hydrocarbon with M+= 132
(b) A hydrocarbon with M+=166
(c) A hydrocarbon with M+= 84
Q 14 P
Question: Propose structures for compounds that fit the following mass-spectral data:
(a) A hydrocarbon with
(b) A hydrocarbon with
(c) A hydrocarbon with
Q15E
From what alkyne might each of the following substances have been made? (Green = Cl.)
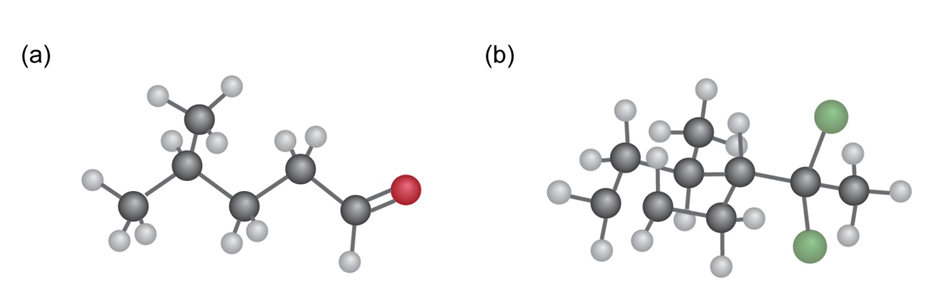
Q15P
Write molecular formulas for compounds that show the following molecularions in their high-resolution mass spectra, assuming that C, H, N,and O might be present. The exact atomic masses are: 1.00783(1H), 12.00000(12C) , 14.00307 (14N) , 15.99491(16O) .
(a) M+ = 98.0844
(b) M+= 123.0320
Q 15 P
Question: Write molecular formulas for compounds that show the following molecular ions in their high-resolution mass spectra, assuming that C, H, N, and O might be present. The exact atomic masses are: 1.00783 , 12.00000 , 14.00307 , 15.99491 .
Q16E
How would you prepare the following substances, starting from any
compounds having four carbons or fewer?