Chapter 12: Q13P (page 354)
Show how you could prepare the following compounds from 4-methyl-3- penten-2-one,
(a)
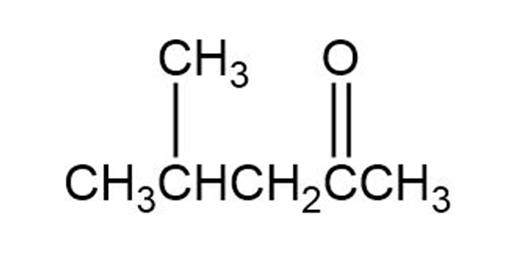
(b)
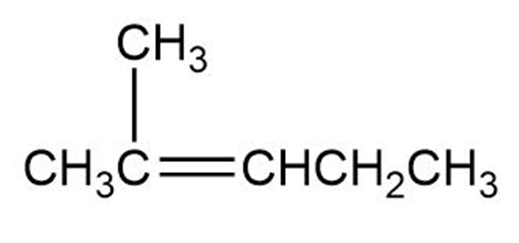
(c)
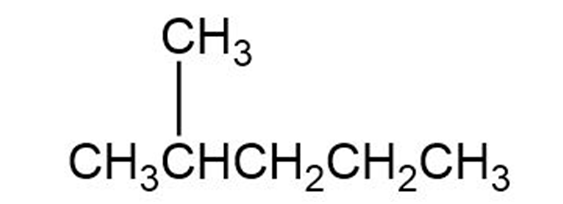
Short Answer
(a)
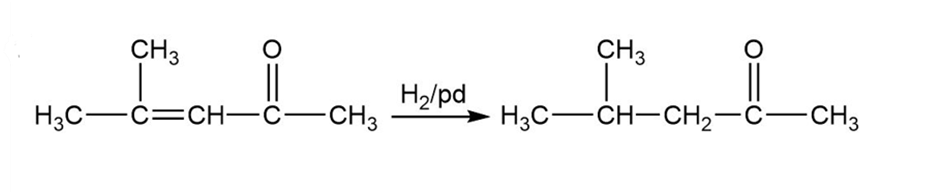
Formation of the desired product
(b)
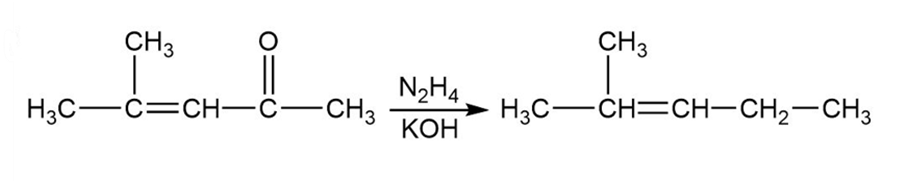
Formation of the desired product
(c)
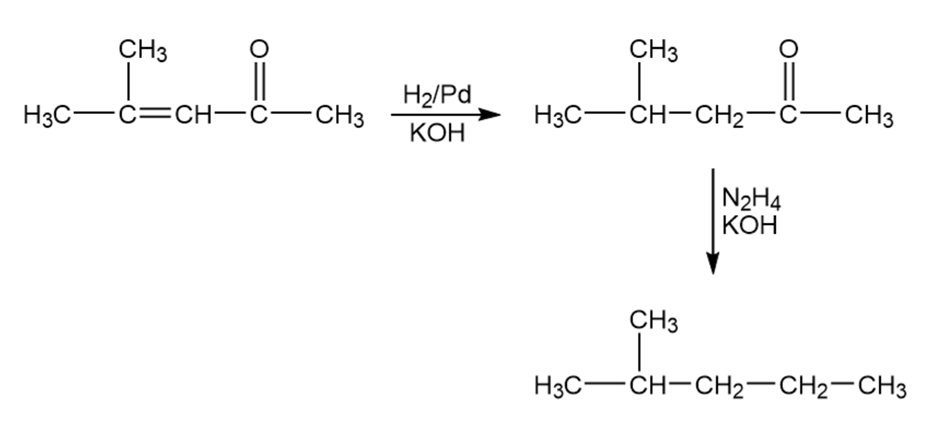
Formation of the desired product
Learning Materials
Features
Discover
Chapter 12: Q13P (page 354)
Show how you could prepare the following compounds from 4-methyl-3- penten-2-one,
(a)

(b)

(c)

(a)

Formation of the desired product
(b)

Formation of the desired product
(c)

Formation of the desired product
All the tools & learning materials you need for study success - in one app.
Get started for free
Camphor, a saturated monoketone from the Asian camphor tree, is used among other things as a moth repellent and as a constituent of embalmingfluid. If camphor has M+=152.1201 by high-resolution mass spectrometry,what is its molecular formula? How many rings does camphor have?
Amino acids can be prepared by reaction of alkyl halides with diethyl acetamidomalonate, followed by heating the initial alkylation product with aqueous HCl. Show how you would prepare alanine, , one of the twenty amino acids found in proteins, and propose a mechanism for acid-catalyzed conversion of the initial alkylation product to the amino acid.

The following two hydrocarbons have been isolated from various plants in the sunflower family. Name them according to IUPAC rules.
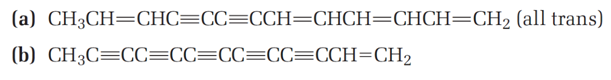
Question: Halogenated compounds are particularly easy to identify by their mass
spectra because both chlorine and bromine occur naturally as mixtures
of two abundant isotopes. Recall that chlorine occurs as 35Cl (75.8%)
and 37C (24.2%); and bromine occurs as 79Br (50.7%) and 81Br (49.3%).
At what masses do the molecular ions occur for the following formulas?
What are the relative percentages of each molecular ion?
(a)Bromomethane, CH3Br
(b)1-Chlorohexane, C6H13Cl
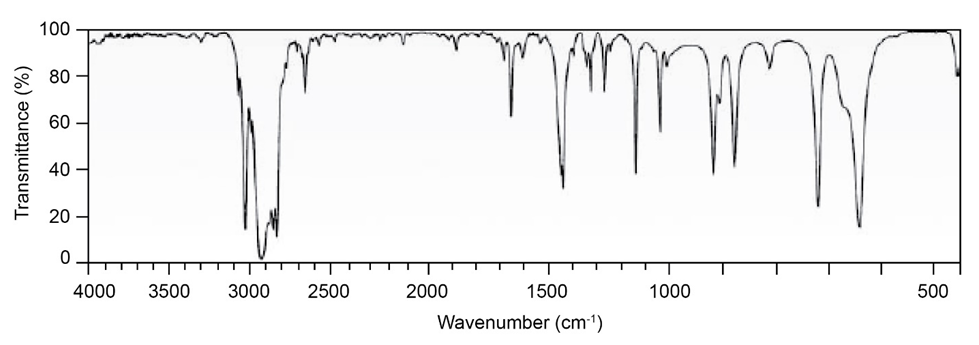
Question: Two infrared spectra are shown. One is the spectrum of cyclohexane,
and the other is the spectrum of cyclohexene. Identify them, and
explain your answer.
(a)
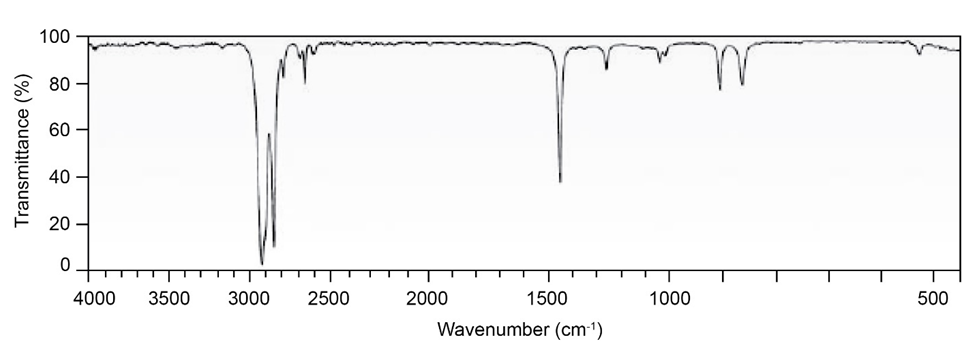
(b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.