Chapter 12: Q 13 P (page 385)
Question: Show the structures of the fragments you would expect in the mass spectra of the following molecules:
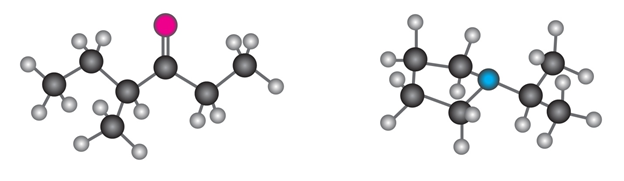
Short Answer
Structure of fragments of molecule:
a)
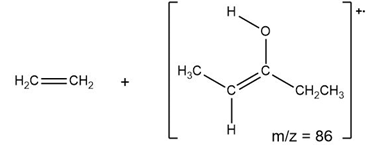
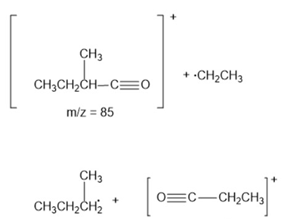
b)
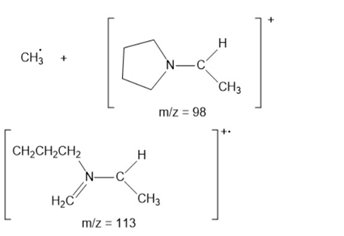
Learning Materials
Features
Discover
Chapter 12: Q 13 P (page 385)
Question: Show the structures of the fragments you would expect in the mass spectra of the following molecules:

Structure of fragments of molecule:
a)


b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Which is stronger, the C=O bond in an ester () or the C=O bond in a saturated ketone ()? Explain
Write molecular formulas for compounds that show the following molecularions in their high-resolution mass spectra, assuming that C, H, N,and O might be present. The exact atomic masses are: 1.00783(1H), 12.00000(12C) , 14.00307 (14N) , 15.99491(16O) .
(a) M+ = 98.0844
(b) M+= 123.0320
Question: How could you use infrared spectroscopy to distinguish between the
following pairs of isomers?
(a)
(b)
Question: The nitrogen ruleof mass spectrometry says that a compound containing an odd number of nitrogen’s has an odd-numbered molecular ion.
Conversely, a compound containing an even number of nitrogen’s has
an even-numbered M+peak. Explain.
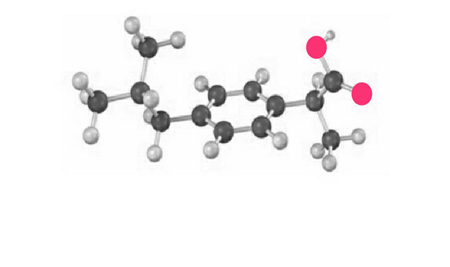
The following model is a representation of ibuprofen, a common over-the-counter pain reliever. Indicate the positions of the multiple bonds, and draw a skeletal structure (gray 5 C, red 5 O, ivory 5 H).
What do you think about this solution?
We value your feedback to improve our textbook solutions.