Chapter 11: Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations
Q 11-11-31 E
We saw in Section 8-7 that bromohydrins are converted into epoxides when treated with base. Propose a mechanism, using curved arrows to show the electron flow.
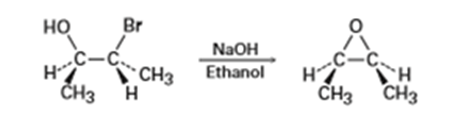
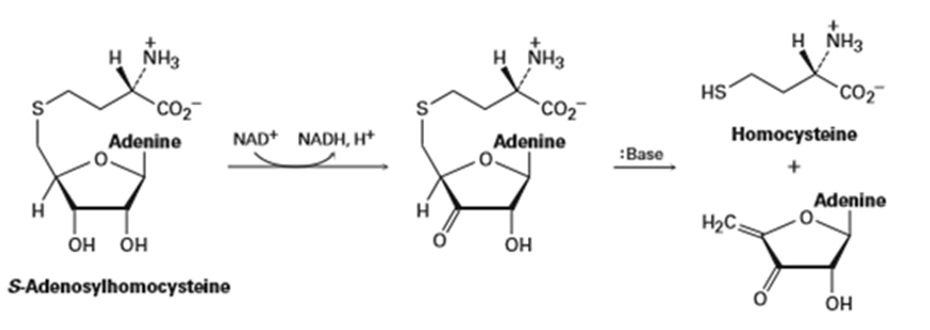
Q 11-11-33 E
Metabolism of S-adenosylhomocysteine (Section 11-6) involves the following sequence. Propose a mechanism for the second step.
Q 11-11-34 E
Reaction of iodoethane withyields a small amount of isonitrile,, along with the nitrileas the major product. Write electron-dot structures for both products, assign formal charges as necessary, and propose mechanisms to account for their formation.
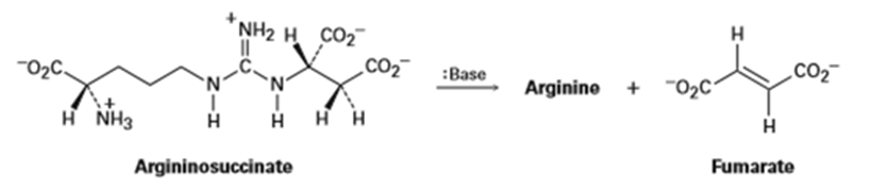
Q 11-11-35 E
One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.
Q 11-11-36 E
Methyl esters () undergo a cleavage reaction to yield carboxylate ions plus iodomethane on heating with LiI in dimethylformamide:
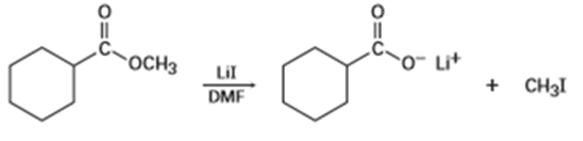
The following evidence has been obtained: (1) The reaction occurs much faster in DMF than in ethanol. (2) The corresponding ethyl ester () cleaves approximately 10 times more slowly than the methyl ester. Propose a mechanism for the reaction. What other kinds of experimental evidence could you gather to support your hypothesis?
Q 11-11-37 E
SN2 reactions take place with inversion of configuration, andSN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of configuration. Propose a mechanism.
.
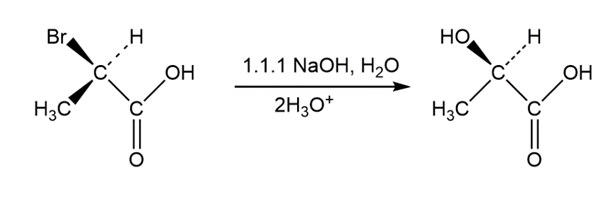
Q 11-11-41 E-a
Which compound in each of the following pairs will react faster in an SN2reaction with?
a) CH3Br or CH3I
Q 11-11-41 E-b
Which compound in each of the following pairs will react faster in an SN2reaction with OH-?
(b) CH3CH2Iin ethanol or dimethyl sulfoxide
Q 11-11-41 E-c
Which compound in each of the following pairs will react faster in an SN2reaction with OH-?
(c)
Q 11-11-41 E-d
Which compound in each of the following pairs will react faster in an SN2reaction with OH-?
(d)
