Chapter 11: Q 11-11-33 E (page 350)
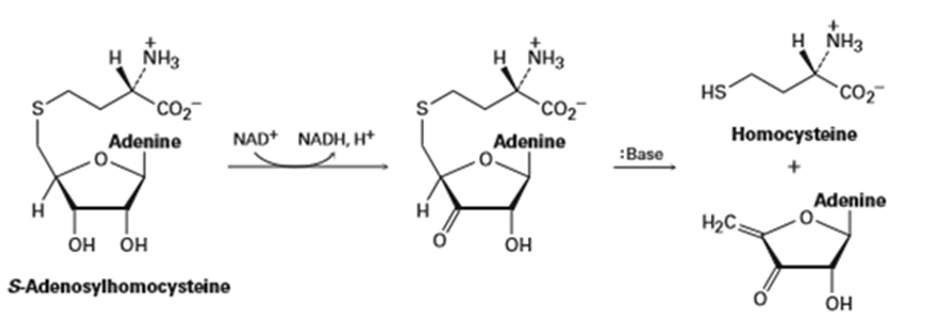
Metabolism of S-adenosylhomocysteine (Section 11-6) involves the following sequence. Propose a mechanism for the second step.
Short Answer
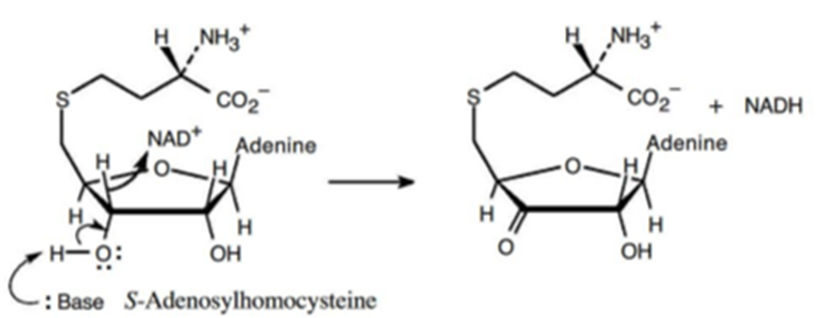
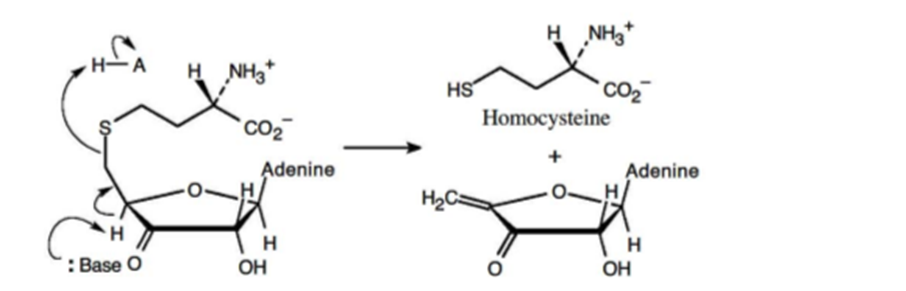
The mechanism for the metabolism of S-adenosylhomocysteine is,
Learning Materials
Features
Discover
Chapter 11: Q 11-11-33 E (page 350)
Metabolism of S-adenosylhomocysteine (Section 11-6) involves the following sequence. Propose a mechanism for the second step.

The mechanism for the metabolism of S-adenosylhomocysteine is,


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: There are eight diastereomers of 1,2,3,4,5,6-hexachlorocyclohexane. Draw each in its more stable chair conformation. One isomer loses HCl in an E2 reaction nearly 1000 times more slowly than the others. Which isomer reacts so slowly, and why?
Which isomer would you expect to undergo E2 elimination faster, trans-1-bromo-4-tert-butylcyclohexane or cis-1-bromo-4-tert-butylcyclohexane? Draw each molecule in its more stable chair conformation, and explain your answer.
3-Bromo-1-butene and 1-bromo-2-butene undergo reaction at nearly the same rate, even though one is a secondary halide and the other is primary. Explain.
What product would you expect from reaction of 1-bromobutane with each of the following?
(a)NaI (b) KOH (c)(d)
Which reactant in each of the following pairs is more nucleophilic? Explain.
b) H2O or CH3CO2-
What do you think about this solution?
We value your feedback to improve our textbook solutions.