Chapter 11: Q 11-11-42 E-b (page 350)
Which reactant in each of the following pairs is more nucleophilic? Explain.
b) H2O or CH3CO2-
Short Answer
CH3CO2-
Learning Materials
Features
Discover
Chapter 11: Q 11-11-42 E-b (page 350)
Which reactant in each of the following pairs is more nucleophilic? Explain.
b) H2O or CH3CO2-
CH3CO2-
All the tools & learning materials you need for study success - in one app.
Get started for free
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
a)
Organic solvents like benzene, ether, and chloroform are neither protic nor strongly polar. What effect would you expect these solvents to have on the reactivity of a nucleophile in reactions?
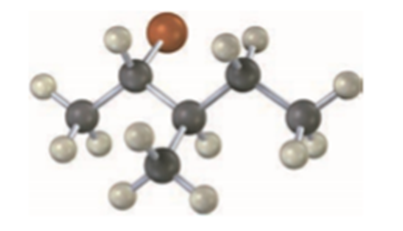
What stereochemistry do you expect for the trisubstituted alkene obtained by E2 elimination of the following alkyl halide on treatment with KOH? (Reddish brown = Br.)
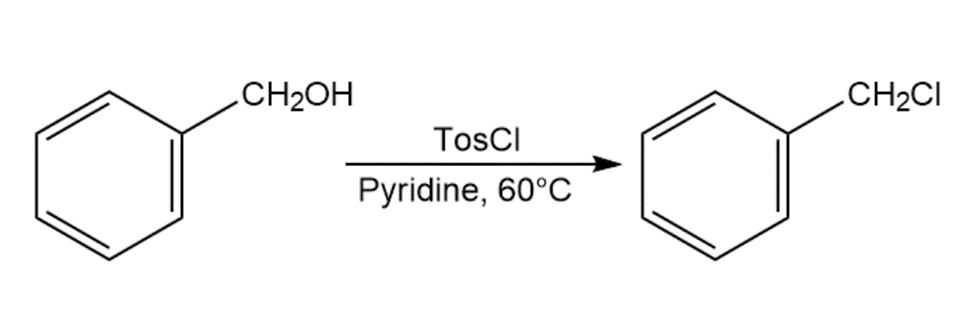
When primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out at a higher temperature, an alkyl chloride is often formed. Explain.
Question:What products would you expect from the reaction of 1-bromopropane with each of the following?
(f) Mg, then H2O
What do you think about this solution?
We value your feedback to improve our textbook solutions.