Chapter 11: Q 11-11-41 E-a (page 350)
Which compound in each of the following pairs will react faster in an SN2reaction with?
a) CH3Br or CH3I
Short Answer
CH3I
Learning Materials
Features
Discover
Chapter 11: Q 11-11-41 E-a (page 350)
Which compound in each of the following pairs will react faster in an SN2reaction with?
a) CH3Br or CH3I
CH3I
All the tools & learning materials you need for study success - in one app.
Get started for free
Compound X is optically inactive and has the formula. On treatment with strong base, X gives hydrocarbon Y,. Compound Y absorbs 2 equivalents of hydrogen when reduced over a palladium catalyst and reacts with ozone to give two fragments. One fragment, Z, is an aldehyde with formula. The other fragment is glyoxal,. Write the reactions involved, and suggest structures for X, Y, and Z. What is the stereochemistry of X?
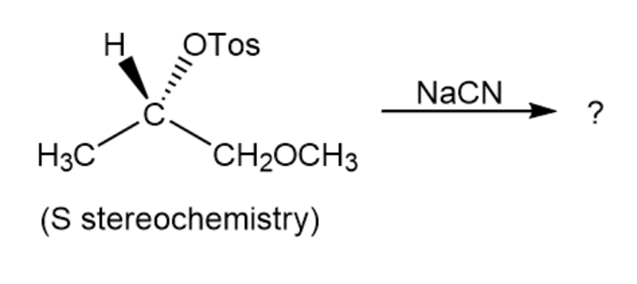
Reaction of the following S to sylate with cyanide ion yields a nitrileproduct that also has S stereochemistry. Explain.
Question: (R)-2-Bromooctane undergoes racemization to give ()-2-bromooctane when treated with NaBr in dimethyl sulfoxide. Explain.
Organic solvents like benzene, ether, and chloroform are neither protic nor strongly polar. What effect would you expect these solvents to have on the reactivity of a nucleophile in reactions?
Which reactant in each of the following pairs is more nucleophilic? Explain.
a) BF3 or F-
What do you think about this solution?
We value your feedback to improve our textbook solutions.